C2h3cl Lewis Structure
What is the following Lewis structure:? 3
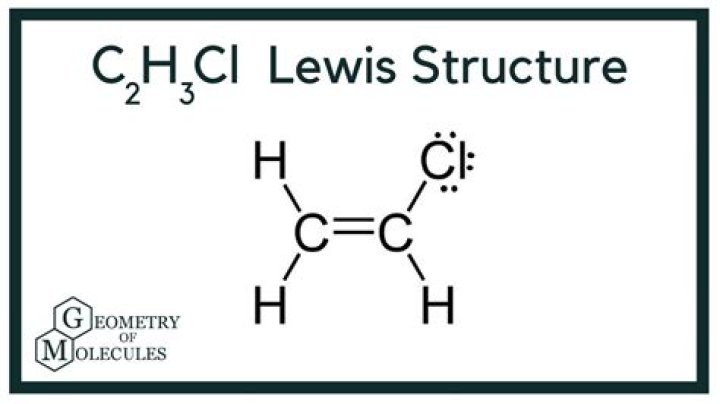
C2H3Cl:
CH2O2:
C2H3N:
CH5N:
C3H6:
HCN:
Lewis structure for C2h3cl
Singing in the upper right corner and moving clockwise we can create 4 resonant structures that will extend the byte P to 10. Overall, the regular charge of P is 0 and so is the oxygen. The final resonance structure increases the octet to 12, but it is not a perfect structure because at least the electronegative P has a more negative formal charge than some O's. Below are four of the best resonant structures. cs2 post bad later.
Working with this framework:
C2H3Cl: H2C = CHCl
CH2O2: HC = O (OH)
C2H3N: H3CCN
CH5N: H3CNH2
C3H6:
HCN: