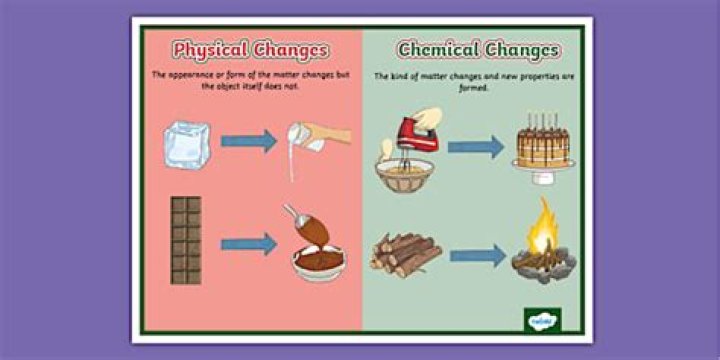
In a physical change the appearance or form of the matter changes but the kind of matter in the substance does not. However in a chemical change, the kind of matter changes and at least one new substance with new properties is formed. … All chemical reactions are reversible although this can be difficult in practice.
What is the difference between physical and chemical change class 9?
1)No new substance is formed in the physical change. A new substance is formed in a chemical change. 2)Physical change is temporary change. A chemical change is a permanent change.
What is the difference between a physical change and a chemical change Lesson 11?
Physical change – A change in the size, shape, color, or state of matter of a substance. No new substance is produced. Chemical change – The change of substances into different substances.
What is an example of both physical and chemical change?
Melting and burning of candle wax is an example of both physical and chemical changes. Answer: Burning of wood is a example of both physical and chemical change. When wood is burnt the moisture present in it turns to vapour ,it is a physical change while it burns and generate CO2 is a chemical change.What is the difference between physical and chemical change Class 10?
When a substance undergoes a physical change, its composition remains the same but there is rearrangement of molecules. … Physical changes are reversible in nature, i.e., original substance can be recovered. Chemical changes are irreversible in nature, i.e., original substance cannot be recovered.
What is the difference between chemical and physical properties?
A physical property is a characteristic of a substance that can be observed or measured without changing the identity of the substance. Physical properties include color, density, hardness, and melting and boiling points. A chemical property describes the ability of a substance to undergo a specific chemical change.
What are 3 differences between physical and chemical changes?
In a physical change, no new substance is formed. A chemical change is always accompanied by one or more new substance(s). Physical change is easily reversible i.e original substance can be recovered. Chemical changes are irreversible i.e. original substance cannot be recovered.
What is the difference between chemical change and chemical reaction?
Originally Answered: What is the difference between a chemical reaction and a chemical change ? In a chemical reaction no atoms are lost. It is basically a mixture of two substances. In a chemical change, a completely new substance with new properties is formed.What do you know about chemical change?
Chemistry. a usually irreversible chemical reaction involving the rearrangement of the atoms of one or more substances and a change in their chemical properties or composition, resulting in the formation of at least one new substance: The formation of rust on iron is a chemical change.
What is the difference between a physical change and a chemical change Lesson 22?In a physical change, no energy is taken in or given off unless there is a change of state. In a chemical change, matter changes from one kind of material to an- other kind of material. The atoms that make up the material do not change. Instead, the atoms change the way they are linked up.
Article first time published onWhat is the difference between physical change and chemical change Grade 5?
chemical changes for kids in 3rd, 4th & 5th grade! In a chemical change, a new substance is made, like when you burn a candle. In a physical change, no new substance is made, like when water turns to ice.
What is the difference between a chemical process and a physical process in chemistry?
There is no real difference between a chemical process and a physical process in chemistry. … According to such teachers, things like burning fuels are chemical processes and things like dissolving salt in water or freezing water into ice are physical processes.
What is the difference between physical and chemical balance?
The key difference between physical and chemical equilibrium is that a physical equilibrium is an equilibrium in which the physical state of the system does not change whereas chemical equilibrium is the equilibrium state in which the concentrations of reactants and products is not changed with time.
Why is it important to know the difference between chemical and physical changes?
It is important to understand the difference between chemical and physical changes. Some changes are obvious, but there are some basic ideas you should know. Physical changes are usually about physical states of matter. … Chemical changes happen when atomic bonds are broken or created during chemical reactions.
How are physical and chemical changes similar and different?
What is the difference between physical and chemical changes? A physical change in a substance doesn’t actually change what the substance is. It remains the same. In a chemical change where there is a chemical reaction, a new substance is formed and energy is either given off or absorbed.
What is difference between physical change and physical property?
All substances have distinct physical and chemical properties, and may undergo physical or chemical changes. Physical properties, such as hardness and boiling point, and physical changes, such as melting or freezing, do not involve a change in the composition of matter.
What is the difference between a physical and chemical property be able to give an example for each property?
So things like colour, melting point, boiling point, density, hardness and so on are all physical properties. Chemical properties can only be measured and observed by making changes to the substance on an atomic/molecular scale by forming or breaking chemical bonds.
What is the basic difference between physical and chemical classification of matter?
Chemical property can only be observed by performing a chemical reaction. When a chemical change happens the molecular structure of a substance changes. Anything that can be measured without changing the identity or chemical composition of substance is known as the physical property of a substance.
What is physical change short answer?
From Simple English Wikipedia, the free encyclopedia. A physical change is a type of change in which the form of matter is altered but one substance is not transformed into another. The size or shape of matter may be changed, but no chemical reaction occurs.
How do you identify a chemical change explain with an example?
Answer: Chemical changes occur when a substance combines with another to form a new substance, called chemical synthesis or, alternatively, chemical decomposition into two or more different substances. … An example of a chemical change is the reaction between sodium and water to produce sodium hydroxide and hydrogen.
What is physical change explain briefly?
What is a physical change? A physical change is a change to the physical—as opposed to chemical—properties of a substance. They are usually reversible. The physical properties of a substance include such characteristics as shape (volume and size), color, texture, flexibility, density, and mass.
What is the difference between physical and chemical change quizlet?
What is the difference between physical and chemical changes? A physical change in a substance doesn’t actually change what the substance is. It remains the same. In a chemical change where there is a chemical reaction, a new substance is formed and energy is either given off or absorbed.
What is physical change in Brainly?
A physical change is a type of change in which the form of matter is altered but one substance is not transformed into another. i.e., the size of shape of the matter may be changed but no chemical reaction occurs. These changes are usually reversible. Muxakara and 11 more users found this answer helpful. Thanks 8.
What is the difference between physical and chemical separation methods?
Physical changes do not alter the nature of the substance, they simply alter the form. Pure substances, such as compounds, can be separated through chemical changes. Chemical changes change the chemical composition of a substance and can only occur through a chemical reaction.
How can you identify a physical change?
- Expected color change.
- Change in size or shape.
- Change in state of matter.
- Reversible.
- No new substance formed!