Converting an alkane hydrocarbon to an alkene involves dehydrogenation, an endothermic process in which hydrogen is removed from the alkane molecule.
How is hydrogen removed from an alkane?
Converting an alkane hydrocarbon to an alkene involves dehydrogenation, an endothermic process in which hydrogen is removed from the alkane molecule.
What happens when alkanes react with hydrogen?
Alkanes (the most basic of all organic compounds) undergo very few reactions. The two reactions of more importaces is combustion and halogenation, (i.e., substitution of a single hydrogen on the alkane for a single halogen) to form a haloalkane.
Do alkanes have hydrogen?
The International Union of Pure and Applied Chemistry (IUPAC) defines alkanes as “acyclic branched or unbranched hydrocarbons having the general formula C nH 2n+2, and therefore consisting entirely of hydrogen atoms and saturated carbon atoms”.Can alkanes form hydrogen bonds?
The simple answer is none, as alkanes do not engage in hydrogen bonding.
What does H2 and Pd do?
Pd/C and hydrogen will reduce alkynes all the way to alkanes – that is, two equivalents of H2 are added. Contrast that to Lindlar’s catalyst, which only adds one equivalent of H2 (but also in syn fashion).
How do you turn an alkane into an alcohol?
Convert the alkane to halogen halide; treat it with magnesium metal and dry ether to form GRIGNERD REAGENT. Then dissolve in water to get the alcohol. The alcohols may be considered as derivatives of alkanes. The replacement of one hydogen atom in an alkane by a hydroxyl (-OH) group gives the corresponding alcohol.
How are hydrogen atoms represented in an alkane skeletal structure?
Note that there is no representation of hydrogens in a skeletal structure. Since, in the absence of double or triple bonds, carbon makes four bonds total, the presence of hydrogens is implicit.How many hydrogen atoms are in the alkane?
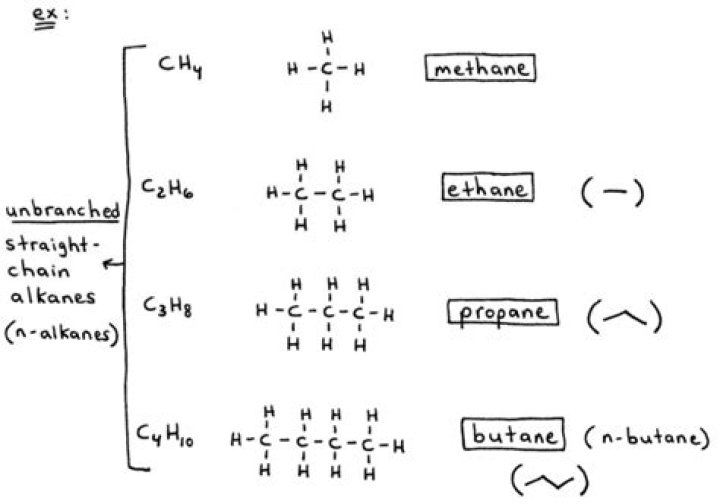
Alkanes have the general formula CnH2n+2. For example, an alkane with 2 (n) carbon atoms, will have 6 (2n + 2) hydrogen atoms. Their adjacent atoms are connected with sigma bonds and form tetrahedral centers around the carbon atoms. As these bonds are all single bonds, there is free rotation around all connections.
How do alkanes and alkenes differ in their molecular structures?While alkanes and alkenes are both hydrocarbons, the primary difference is that alkanes are saturated molecules, containing only single covalent bonds (σ-bonds) between the carbon atoms whereas alkenes are unsaturated molecules containing a double covalent bond (combination of a π-bond and a σ-bond).
Article first time published onCan you oxidize an alkane?
Alkanes can be oxidized to carbon dioxide and water via a free‐radical mechanism. The energy released when an alkane is completely oxidized is called the heat of combustion. … The lower the potential energy of a compound, the more stable it is.
How do alkanes react?
Alkanes undergo a substitution reaction with halogens in the presence of light. For instance, in ultraviolet light , methane reacts with halogen molecules such as chlorine and bromine. This reaction is a substitution reaction because one of the hydrogen atoms from the methane is replaced by a bromine atom.
How do you add bromine to an alkane?
Unsaturated hydrocarbons such as alkenes and alkynes are much more reactive than the parent alkanes. They react rapidly with bromine, for example, to add a Br2 molecule across the C=C double bond. This reaction provides a way to test for alkenes or alkynes.
How do you make an alkane?
Preparation of Alkanes from unsaturated hydrocarbon: Alkane can be prepared from alkene and alkyne through the process of hydrogenation. In this process, dihydrogen gas is added to alkynes and alkenes in the present catalyst. This catalysts which are finely divided is like nickel, palladium or platinum to form alkanes.
Can alkenes hydrogen bond?
An example of an alkene addition reaction is a process called hydrogenation.In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane. … A hydrogen atom is then transferred to the alkene, forming a new C-H bond.
Can alkenes have hydrogen bonds?
Alkenes are nonpolar, since they contain nothing but carbon-carbon and carbon-hydrogen bonds, and are not soluble in water; they are also generally less dense than water. Many alkenes are isolated from petroleum, and may serve as the starting points for the synthesis of more complex molecules.
How do you go from an alkane to an alkyne?
Dehydrohalogenation. The loss of a hydrogen atom and a halogen atom from adjacent alkane carbon atoms leads to the formation of an alkene. The loss of additional hydrogen and halogen atoms from the double‐bonded carbon atoms leads to alkyne formation.
How do you make a Haloalkane?
Halogenoalkanes can be made from the reaction between alkenes and hydrogen halides, but they are more commonly made by replacing the -OH group in an alcohol by a halogen atom. That’s the method we’ll concentrate on in this page.
How do you convert alkane to Haloalkane?
The reaction of a halogen with an alkane in the presence of ultraviolet (UV) light or heat leads to the formation of a haloalkane (alkyl halide). An example is the chlorination of methane.
Can H2 reduce ketone?
H2 Ni are strong reducing agents which reduces aldehydes to primary alcohols,ketones to secondary alcohols and carboxylic acid and its derivatives to primary alcohols.
How do you remove palladium from carbon?
Purely inorganic Palladium salts can be removed by liquid extraction of a solid residue (or the organic compounds are extracted otherwise). A better separation is achieved by liquid-liquid extraction. In this case the polarity of the aqueous ssolution can be increased by Addition of sodium chloride. Or use edta, etc.
Can H2 reduce esters?
The reduction of carboxylic acid esters to alcohols is commonly effected with a stoichiometric amount of a highly reactive metal-hydride reagent (e.g. LiAlH4). The amount of waste generated by this procedure would decrease strongly through the use of H2 as the reducing agent.
What alkane contains 28 hydrogen atoms?
Number of C atomsNumber of isomersMolecular Formula27240215803C27H5628617105614C28H58291590507121C29H60304111846763C30H62
How many bonds does an alkane have?
Alkanes. Alkanes, or saturated hydrocarbons, contain only single covalent bonds between carbon atoms. Each of the carbon atoms in an alkane has sp3 hybrid orbitals and is bonded to four other atoms, each of which is either carbon or hydrogen.
How many hydrogen atoms are in an acyclic alkane with carbon atoms?
We have the general formula of alkane as CnH2n+2 . Hence, there are 32 hydrogen atoms present in an acyclic alkane with 15 carbon atoms.
When one hydrogen atom of alkane is removed then it is called?
An alkyl group is a group of atoms that results when one hydrogen atom is removed from an alkane.
How many hydrogen atoms are in an alkane with 6 carbon atoms?
Each carbon atom in this normal alkane, where n = 6, has at least two hydrogen atoms bonded to it, which accounts for the 2n in the general formula. Each of the two terminal carbon atoms has another hydrogen atom bonded to it, which accounts for the +2 in the subscript on hydrogen in the general formula.
What happens when an alkane boils?
The boiling points of alkanes increase with increasing number of carbons. This is because the intermolecular attractive forces, although individually weak, become cumulatively more significant as the number of atoms and electrons in the molecule increases.
How is the ratio of carbon to hydrogen different for alkanes alkenes and alkynes?
The saturated alkane has the highest ratio of hydrogen to carbon. The unsaturated alkene and the six membered alkane ring share the same chemical formula. It is important to remember this relationship. The unsaturated alkyne has a lower ratio of hydrogen to carbon than alkenes with a second pi bond.
What is the difference in the number of hydrogen atoms between alkynes and alkanes?
Alkynes are hydrocarbons (compounds containing only C and H) that have one or more triple bonds (two C atoms are joined by 6 shared electrons). The molecular formula fits the formula CnH2n-2 and has therefore four hydrogen atoms less than the corresponding alkane.
What is pyrolysis of alkane?
Pyrolysis is defined as the conversion of a compound into smaller fragments in the absence of air through the application of heat. It is different from combustion. It happens in the absence of air and hence oxidation of compounds does not take place. Generally, pyrolysis of alkanes is also named as cracking.