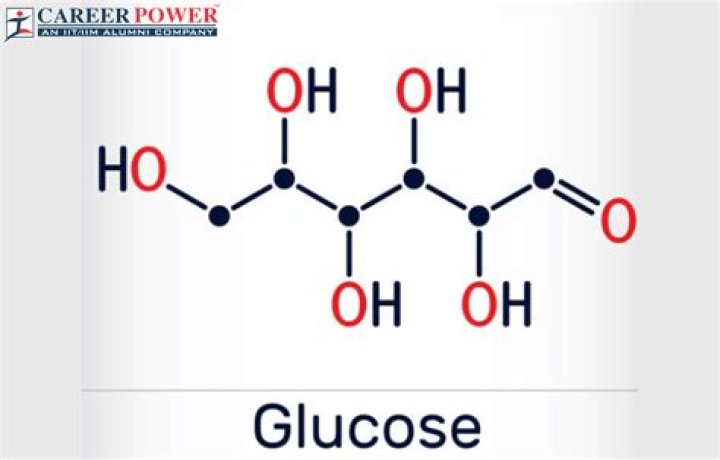
4 Simple Steps – Chain Glucose Molecule. … Draw 6 carbon molecules and draw arms except for the first one.Draw 5 hydrogen to carbon bonds. … Fill remaining spaces with ( OH ) group. … Complete the top end with hydrogen bonds and 1 oxygen double bond.9 thoughts on “4 Simple Steps – Chain Glucose Molecule”
How do you write glucose?
Formulas. The molecular formula for glucose is C6H12O6. This means that there are 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms bonded together to make one molecule of glucose.
How does glucose have 2 isomers?
Chain and Ring forms of Glucose When glucose forms the ring structure, it can form two isomers. … The α and β forms interconvert in a period of a few hours in water solution, however when glucose polymerises, the two forms make polymers with very different properties (starch and cellulose, see later).
What are the two types of glucose?
Glucose occurs naturally in two distinct types of molecular arrangements known as L-glucose and D-glucose isomers. These glucose isomers both contain identical molecules but are arranged in a mirror reflection of each other.What is the difference between α glucose and β glucose?
The main difference between alpha and beta glucose is that the –OH group attached to the first carbon atom in alpha glucose is located on the same side as the –CH2OH group whereas the –OH group attached to the first carbon atom of in beta glucose is located on the opposite side from the –CH2OH group.
Is beta glucose the same as galactose?
Compare Glucose and Galactose in the Chair Structures Galactose is defined as the -OH on C # 4 in a upward projection in the chair form,(also upward in the Haworth structure). Both glucose and galactose may be either alpha or beta on the anomeric carbon, so this is not distinctive between them.
How do I know if my sugar is alpha or beta?
α (Alpha) – the name given to the configuration of a cyclic sugar where the oxygen on the anomeric carbon is on the opposite face of the ring relative to the substituent on the other carbon flanking the ring oxygen. Contrasted with beta (β) which is where the two substituents are on the same faces of the ring.
What does C6H12O6 look like?
C6H12O6GlucoseDensity1.54 g/cm³Melting Point146 °CSimple sugarMonosaccharideWhy is glucose Dextrorotatory?
Glucose is by far the most common carbohydrate and classified as a monosaccharide, an aldose, a hexose, and is a reducing sugar. It is also known as dextrose, because it is dextrorotatory (meaning that as an optical isomer is rotates plane polarized light to the right and also an origin for the D designation.
What is glucose a level biology?Introduction. Glucose is the most important monosaccharide present in our body. It belongs to the hexose category of monosaccharides. Glucose provides energy to all the cells in our body except the cardiac myocytes. Excess glucose is stored in the body in the form of storage molecules.
Article first time published onWhat is Alpha glucose?
Starch: Alpha glucose is the monomer unit in. starch. As a result of the bond angles in the alpha acetal linkage, starch (amylose) actually forms a spiral structure.
Where is glucose most commonly found?
glucose, also called dextrose, one of a group of carbohydrates known as simple sugars (monosaccharides). Glucose (from Greek glykys; “sweet”) has the molecular formula C6H12O6. It is found in fruits and honey and is the major free sugar circulating in the blood of higher animals.
What type of sugar is glucose?
Glucose is a simple sugar or monosaccharide. It’s your body’s preferred carb-based energy source ( 1 ). Monosaccharides are made up of one single unit of sugar and thus cannot be broken down into simpler compounds. They’re the building blocks of carbohydrates.
What is the most common form of glucose?
The most common form of glucose is d-glucose.
How do you find isomers of glucose?
We see that carbon atoms 2, 3, 4, and 5 are chiral centres. The maximum number of optical isomers is 2n , where n is the number of chiral centres. In glucose, n=4 , so 24 = 16 possible isomers.
What are the isomers of glucose called?
Glucose and its isomers Fructose is a structural isomer of glucose and galactose, meaning that its atoms are actually bonded together in a different order. Glucose and galactose are stereoisomers (have atoms bonded together in the same order, but differently arranged in space).
Why is glucose an isomer?
Glucose, galactose, and fructose are monosaccharide isomers, which means they all have the same chemical formula but differ structurally and chemically.
Why is it called alpha and beta glucose?
Alpha glucose is an isomer of glucose that has –OH group present on the first carbon atom is on the same side as that of the CH2OH molecule group. Beta glucose is also an isomer of D-glucose in which the -OH group placed on the first carbon atom is placed on the opposite side of the CH2OH group.
How can you tell the difference between D and L glucose?
* L-glucose: D-glucose and L-glucose are made up of the same atoms. The only difference between the two structures is displayed through fischer projection. Unlike D-glucose, the oxygen and hydrogen group of atoms in L-glucose points to the left in fischer projection.
Why is there alpha and beta glucose?
Alpha and beta glucose form the basis for two key components of a healthy diet: starch and fiber. Glucose is one of your body’s favorite sources of energy. Alpha and beta glucose are two common forms of the fuel that differ in their molecular structure and the types of nutrients they create.
What is beta glucose?
Beta-glucose is a cyclic, six carbon sugar, formed from glucose, and can polymerise to make cellulose, an essential polysaccharide used in the structure of plants. … These two isomers exist because the formation of cyclic glucose creates an asymmetric carbon at C1.
What sugars make lactose?
Lactose is a sugar found in milk. It is a disaccharide made up of glucose and galactose units. It is broken down into the two parts by an enzyme called lactase. Once broken down, the simple sugars can be absorbed into the bloodstream.
What is a furanose ring?
The furanose ring is a cyclic hemiacetal of an aldopentose or a cyclic hemiketal of a ketohexose. A furanose ring structure consists of four carbon and one oxygen atom with the anomeric carbon to the right of the oxygen. … It is the opposite in an l-configuration furanose.
What is the difference between glucose and mannose?
The key difference between glucose galactose and mannose is that glucose is a six-carbon structure and galactose is the C4 epimer of glucose, whereas mannose is the C2 epimer of glucose. … For example, galactose and mannose are epimers of glucose.
Is galactose ad sugar or an L sugar?
Identifiersshow SMILESPropertiesChemical formulaC6H12O6Molar mass180.156 g·mol−1
What is difference between galactose and glucose?
Galactose is the isomer of glucose. They differ only in the organization of their atoms. Glucose and galactose are stereoisomers of each other. The main structural difference in between galactose and glucose is the orientation of the hydroxyl group (OH) at carbon 4.
Is glucose a laxative?
l-Glucose was also found to be a laxative, and has been proposed as a colon-cleansing agent which would not produce the disruption of fluid and electrolyte levels associated with the significant liquid quantities of bad-tasting osmotic laxatives conventionally used in preparation for colonoscopy.
Is D-glucose always Dextrorotatory?
D-compounds are always dextro and L-compounds are always laevorotatory.
What is Dextrorotatory glucose?
Dextrose refers to the dextrorotatory isomer of glucose. By dextrorotatory, it means that it is capable of rotating the plane polarized light in the clockwise direction. … Dextrose and levulose are obtained by the inversion of cane sugar or sucrose, and hence called invert sugar.
What biomolecule is glucose?
Glucose, a 6-carbon sugar, is a simple carbohydrate or “mono-saccharide.” Sugar is a source of quick energy for the body because it is easily metabolized (broken down). Larger, more “complex carbohydrates” are made by stringing together chains of glucose subunits into di-saccharides, tri-saccharides, poly-saccharides.
What are the elements that make up glucose?
This molecule of the sugar glucose consists of 6 carbon atoms bonded together as a chain with additional atoms of oxygen and hydrogen.