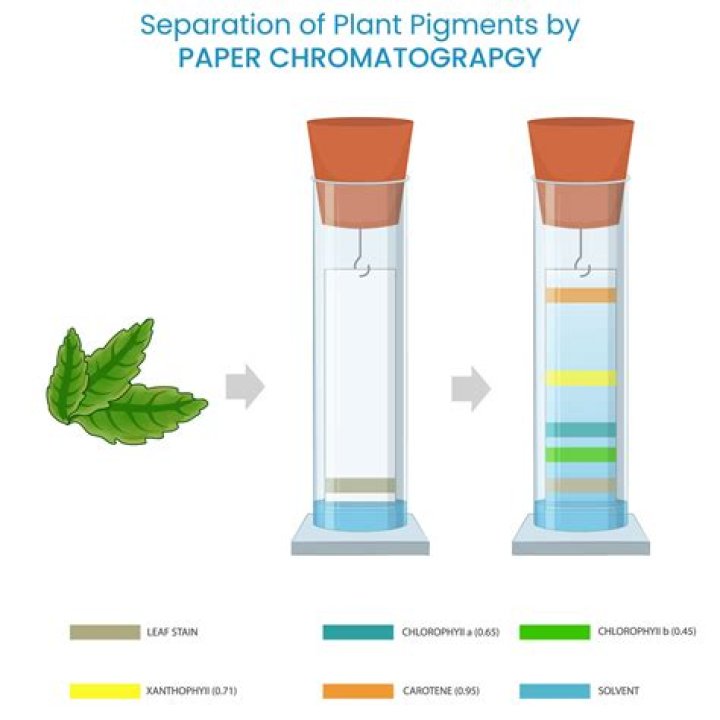
A mixture of unknown amino acids can be separated and identified by means of paper chromatography. The position of the amino acids in the chromatogram can be detected by spraying with ninhydrin, which reacts with amino acids to yield highly coloured products (purple).
How can chromatography be used to identify amino acids?
The most common use is to separate amino acids from a liquid and each other. A spot of the sample is placed on a sheet of glass treated with an absorbent substance. The glass is then placed in a solvent that will travel up the absorbent surface and cause the solid to move out of the liquid with it.
Which is the reagent used to identify amino acid in paper chromatogram?
Paper Chromatography Ninhydrin (or fluorescamine) is very useful in chromatographic methods for the analysis of amino acids.
How are the amino acid visualized on the chromatography paper?
Mixtures of amino acids can be separated on chromatographic paper. The separated amino acids are visualized using solution of ninhydrin. Purple color develops upon reaction of amino acid with ninhydrin.How can amino acids be identified?
The amino acid sequence of a protein is determined by the information found in the cellular genetic code. … These gene codes not only determine the order of amino acids in a protein, but they also determine a protein’s structure and function.
Can paper chromatography be used to separate and identify a very volatile substance?
Volatile or complex substances cannot be separated using paper chromatography techniques. The method is not useful for testing a large number of samples and data is not as accurate as in the case of the TLC technique.
How do you identify unknown amino acids?
Due to the nature of amino acids, a titration curve can be employed to identify an unknown amino acid. A titration curve is the plot of the pH versus the volume of titrant used. In the case of amino acids, the titrant will be both an acid and a base.
How can TLC identify unknown amino acids?
Since amino acids are colourless compounds, ninhydrin is used for detecting them. To identify this, after development, the TLC plate is sprayed with ninhydrin reagent and dried in an oven, at 105°C for about 5 minutes. … Rf values can be calculated and compared with the reference values to identify the amino acids.How are the amino acids made visible in the final chromatogram?
Amino acids are colorless compounds. In order to see the spots on the chromatogram, you will apply a solution of ninhydrin to the paper. Ninhydrin will react with the amino acid to produce a purple compound. … Like the amino acids, the medicine tablet ingredients will not be visible after the plate has been developed.
Which amino acid traveled higher on the chromatography paper?Which amino acid traveled higher on the chromatography paper? Explain your reasoning. Since the Rf value is calculated as distance spot moved/distance solvent moved, the higher the Rf value, the further that spot has moved. Leucine has traveled higher.
Article first time published onWhat is paper chromatography of amino acid?
Paper chromatography is especially useful in characterizing amino acids. The different amino acids move at differing rates on the paper because of differences in their R groups. … Rf is simply the distance the biomolecule moved through the filter paper divided by the distance the solvent moved through the paper.
What reagent is paper chromatography?
FOR the paper chromatography of steroids several colour reagents are used, such as 2,4-dinitrophenyl-hydrazine, Zimmermann’s reagent, phosphomolybdic acid, antimony trichloride, etc. Their common defect is that the reagent gives some colour with the paper itself, and so the contrast between spot and paper is too small.
How do you classify amino acids as acidic or basic?
Since an amino acid has both an amine and acid group which have been neutralized in the zwitterion, the amino acid is neutral unless there is an extra acid or base on the side chain. If neither is present then then the whole amino acid is neutral.
How do you identify an amino acid side chain?
The Basic Structure of an Amino Acid The amino acid has a central chiral carbon called the alpha carbon (black). Attached to the central carbon you have a hydrogen atom (gray), an amino or NH2 group (green), and a carboxylic acid COOH group (purple). Finally we have the R group (red), which is a variable side chain.
How can a titration identify an amino acid?
The pKa-values of the amino acid are determined from the full titration graph. To determine pKa1 and pKa2, locate the volume on the graphs half way between the two equivalence point volumes determined from the expanded derivative curves. The pH at this point is in the titration is equal to pKa2.
How do you test for amino acids in the laboratory?
Using a toothpick or a capillary tube, apply a small spot of each amino acid solution to the marked spots on the paper. You will test phenylalanine, alanine, glutamic acid, serine, lysine, aspartic acid, and an unknown amino acid. Use a different applicator for each amino acid and be careful not to mix them up.
What defines an amino acid?
Amino acids are molecules that combine to form proteins. Amino acids and proteins are the building blocks of life. When proteins are digested or broken down, amino acids are left. The human body uses amino acids to make proteins to help the body: Break down food.
What do you know about paper chromatography?
paper chromatography, in analytical chemistry, technique for separating dissolved chemical substances by taking advantage of their different rates of migration across sheets of paper. … The method consists of applying the test solution or sample as a spot near one corner of a sheet of filter paper.
What type of chromatography is paper chromatography?
Answer: Paper chromatography is a type of partition chromatography.
Why do we need to stain amino acids in paper chromatography?
Amino acids can be identified and separated using paper chromatography, a cheaper alternative to using TLC. The amino acids need to be stained with ninhydrin to reveal the spots after the run.
How do you read TLC results?
In simple terms, this value is an indication of how far up a TLC-plate a compound has wandered. A high Rf -value indicates that the compound has travelled far up the plate and is less polar, while a lower Rf -value indicates that the compound has not travelled far, and is more polar.
Which reagent is used as a spraying reagent to detect amino acids on TLC plates?
A new spray reagent, 2-hydroxy-1-naphthaldehyde has been introduced for the detection of amino acids, which is capable of developing several distinguishable colors with high sensitivity (0.06–2.0 μg) on TLC plates.
What factors are going to determine how far a particular amino acid travels up the chromatography paper?
The solvent moves up the paper and over the solutes. The individual solutes will be partitioned between the stationary and mobile phases according to their relative solubility in each phase. The more soluble the solute in the nonpolar, organic, mobile phase, the farther it will move.
Which solution is used as developer in the chromatography of amino acid?
Since amino acids are colourless compounds, ninhydrin is used for detecting them.
What are the RF values of amino acids?
Amino acidRf valueisoleucine0.72leucine0.73lysine0.14methionine0.55
What does paper chromatography separate?
Chromatography can be used to separate mixtures of coloured compounds . Mixtures that are suitable for separation by chromatography include inks, dyes and colouring agents in food.
What are amino acids classify them with examples?
Amino acids are the best-known examples of zwitterions. They contain an amine group (basic) and a carboxylic group (acidic). The -NH2 group is the stronger base, and so it picks up H+ from the -COOH group to leave a zwitterion. The (neutral) zwitterion is the usual form of amino acids that exist in the solution.
How do you identify C and N terminus?
A peptide has two ends: the end with a free amino group is called the N-terminal amino acid residue. The end with a free carboxyl group is called the C-terminal amino acid residue.
What are the different separation and identification methods for amino acids?
Chromatography methods based on partition are very effective on separation, and identification of small molecules as amino acids, carbohydrates, and fatty acids. However, affinity chromatographies (ie. ion-exchange chromatography) are more effective in the separation of macromolecules as nucleic acids, and proteins.
How can you tell if an amino acid is hydrophilic or hydrophobic?
Moreover, hydrophobic amino acids have long side chains with mostly carbon and hydrogen atoms whereas hydrophilic amino acids have either short side chains or side chain with hydrophilic groups.
How do you classify amino acids as polar and nonpolar?
Polar amino acids are hydrophilic. Nonpolar amino acids are hydrophobic. Examples of polar amino acids include serine, lysine and aspartic acid. Alanine, valine, leucine, isoleucine, phenylalanine, glycine, tryptophan, methionine, proline.