General corrosion occurs when most or all of the atoms on the same metal surface are oxidized, damaging the entire surface. Most metals are easily oxidized: they tend to lose electrons to oxygen (and other substances) in the air or in water. As oxygen is reduced (gains electrons), it forms an oxide with the metal.
What are the conditions for a corrosion to occur?
Three things are necessary for corrosion to occur: an electrolyte, an exposed metal surface, and an electron acceptor. Corrosion can be prevented by removing one of these conditions. Coating a metal surface with paint or enamel provides a barrier between the metal and the moisture in the environment.
What are the main causes of corrosion?
Too much humidity or condensation of water vapour on metal surfaces are the primary causes of corrosion. Corrosive gases such as chlorine, hydrogen oxides, ammonia, sulfur oxides, amongst others can result in corrosion of parts of electronic equipment, etc. Corrosion can also occur due to hydrogen and oxygen exposure.
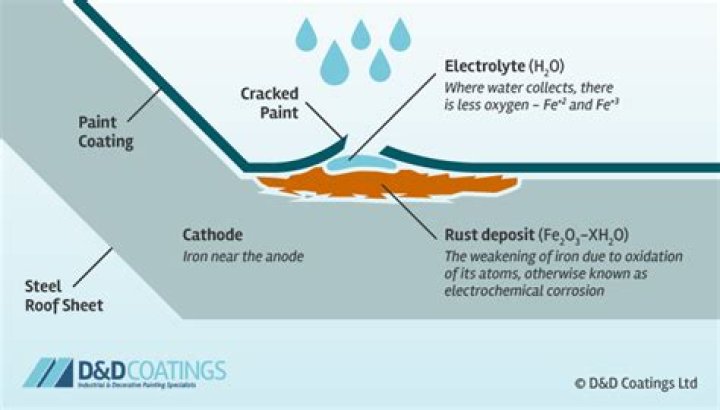
How does corrosion occur in metal iron?
Iron corrodes (forms rust) when exposed to water and oxygen. The rust that forms on iron metal flakes off, exposing fresh metal, which also corrodes. One way to prevent, or slow, corrosion is by coating the metal. Coating prevents water and oxygen from contacting the metal.Does corrosion only occur in metals?
Corrosion is defined as an attack on a material as a result of chemical, frequently electrochemical reaction, with the surrounding medium. According to this definition, the term corrosion can be applied to all materials, including non-metals.
What are the effects of corrosion on metals?
Corrosion also degrades important infrastructure such as steel-reinforced- highways, electrical towers, parking structures and bridges. In short, corrosion is a subject that bears further investigation, so that you can understand how this hidden degradation impacts your life.
How does corrosion of metals affect conductivity?
Corrosion decreases the electrical conductivity of the material and the pH level of sulfuric acid, consequently making the material dull and coarse. … Despite the corrosion problems it creates, metal use is low cost, easy to fabricate, light, and has good heat and electric conductivity.
What is corrosion basically?
Answer. 62.7k+ views. Hint: Corrosion is when a refined metal is naturally converted to a more stable form such as its oxide, hydroxide or sulphide state this leads to deterioration of the material. It can also be considered as eating metals by air.Which metal will corrode fastest?
Also, iron looks like its most affected and also fastest to corrode because out of all those metals, iron is really the only one that rusts.
Which metal is highly corrosive?In these following options, Zinc is a highly corrosive metal. The corrosion of Zinc occurs when it is exposed to moisture and air. Then, a thin oxide layer is formed after the reaction between zinc and oxygen.
Article first time published onWhat causes corrosion in pipes?
Common issues that cause corrosion include a high level of dissolved oxygen (called oxygen corrosion), pipes that were not installed correctly, improper electrical grounding, high flow rates, and a lot of sediment in the water.
What is corrosion of metal in chemistry?
Corrosion is a natural process that converts a refined metal into a more chemically stable form such as oxide, hydroxide, carbonate or sulfide. It is the gradual destruction of materials (usually a metal) by chemical and/or electrochemical reaction with their environment.
Which two metals dont corrode easily?
Gold and Platinum are the metals which do not corrode easily.
Why does corroding reduce conductivity?
Electrical conductivity will decrease as rust or iron oxide can contain moisture and becomes a poor conductor. …
How does steel corrode?
Steel corrosion involves an electrolysis in which the metallic surface releases electrons into an electrolyte, such as a layer of moisture in the presence of oxygen. … Steel can be corroded by the chemical reaction of certain chemicals like sulfuric acid.
What is oxidation corrosion?
22.5. Oxidation is defined as a corrosion reaction in which the corroded metal forms an oxide. Usually, this reaction is with a gas containing oxygen, such as air. Metal systems need to form dense, well-adherent oxide films. The oxidation of steel does not form well-adherent oxide scales.
How are noble metals different from most other metals?
The noble metals are a group of metals that resist oxidation and corrosion in moist air. The noble metals are not easily attacked by acids. They are the opposite of the base metals, which more readily oxidize and corrode.
What is corrosion explain its effect?
Corrosion Definition: Corrosion (from the Latin word corrodes, meaning “to gnaw”) is the irreversible damage or destruction of living tissue or material due to a chemical or electrochemical reaction. Example: A prime example of corrosion is rusting of iron or steel.
What is the mechanism of corrosion?
Oxidation reaction takes place on the surface of the metal, when it is exposed to moisture. A reduction takes place on low energy area of the metal surface. When oxidation and reduction takes place at respective anode and cathode simultaneously and hence corrosion takes place.
Does Aluminium get rust?
Can Aluminium Rust? Aluminium doesn’t rust. But, it’s essential also to note that aluminium in its pure form, is a highly reactive metal. Technically, pure aluminium dissolves when exposed to water but, it’s reactive nature could also be its greatest asset.
Is glass corrosive?
Glass has a high degree of both corrosion and chemical resistance. For this reason it is the primary packaging material to preserve medicines and chemicals. However, over time at high humidity and pH, some glass types corrode. Corroded glass loses its transparency and its strength is reduced.
How can corrosion be prevented?
- Use non-corrosive metals, such as stainless steel or aluminium.
- Make sure the metal surface stays clean and dry.
- Use drying agents.
- Use a coating or barrier product such as grease, oil, paint or carbon fibre coating.
- Lay a layer of backfill, for example limestone, with underground piping.
What are the two main factors that affect corrosion?
- More the reactivity of metal, the more will be the possibility of the metal getting corroded.
- The impurities help in setting up voltaic cells, which increase the speed of corrosion.
- Presence of electrolytes in water also increases the rate of corrosion.
What is rusting How can we prevent rusting?
Rust is an iron oxide(usually red oxide) formed by redox reaction of iron and oxygen in the presence of moisture.. … Rust can be prevented by using rust resistant alloys , galvanisation, cathodic protection , coatings and painting , bluing , by using inhibitors and even by humidity control.
Which metal acts as a sacrificial anode for iron articles?
The addition of zinc, a sacrificial anode, would prevent the iron metal from “corroding”.
Why do metals not corrode easily?
Answer : Metals which have low reactivity are not affected by air, moisture or ordinary chemicals,therefore, they do not corrode easily.
How is corrosion different from rusting?
Corrosion is the process by which certain materials, metals and non-metals, deteriorate as a result of oxidation. Rusting is oxidation of iron in the presence of air and moisture. Corrosion can occur on materials such as ceramics or polymers. Rusting occurs on surfaces of iron and its alloys.
What metals dont oxidize?
In their natural form, noble metals, such as platinum or gold, resist oxidation. Ruthenium, rhodium, palladium, copper, osmium, and iridium are other metals of this type. Humans also invented many corrosion-resistant alloys, such as stainless steel and brass.
What causes copper to corrode?
The presence of oxidizing acids; heavy-metal salts, sulfur, and ammonia; and a number of sulfur and ammonia compounds can cause corrosion to set in. Water that comes from a well is much more likely to contain these materials and put copper lines in jeopardy—but it can occur in the civic water system as well.
Why does corrosion occur in steel pipe connected to copper plumbing?
6. Why does corrosion occur in steel pipe connected to copper plumbing? Hints: Steel is an alloy of iron, which is higher in the electrochemical series. … Hence, iron undergoes oxidation and gets corroded when steel pipe is connected to copper plumbing is exposed to a corroding environment.
How does copper get corroded?
Copper corrosion is the corrosion of materials made of copper or copper alloys. When exposed to the atmosphere, copper oxidizes, causing normally bright copper surfaces to tarnish. After a few years, this tarnish gradually changes to dark brown or black, and finally to green. … This leaching is a product of corrosion.