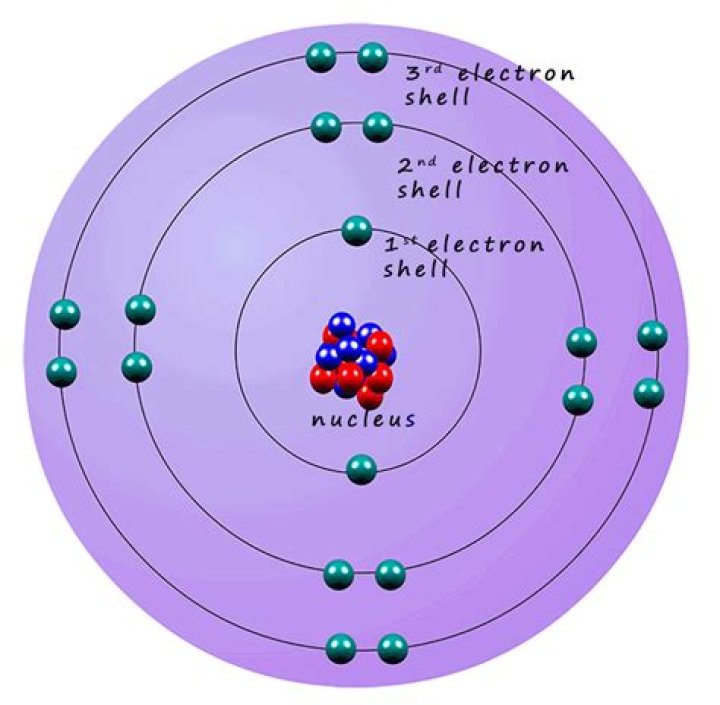
So… for the element of LITHIUM, you already know that the atomic number tells you the number of electrons. That means there are 3 electrons in a lithium atom.
How many electrons does the neutral atom have lithium?
Hence, neutral lithium atoms must contain 3 electrons.
How many electrons are in a neutral atom of lithium gizmo?
Create a lithium atom (3 protons, 4 neutrons, 3 electrons). 4. Diagram: Turn on Show electron dot diagram. The valence electrons of an atom are shown in an electron dot diagram.
How many electrons are in a neutral atom of lithium quizlet?
A lithium has 3 protons, 4 neutrons, and 3 electrons.How many valence electrons does a neutral atom of lithium have?
Lithium has a single electron in the second principal energy level and so we say that lithium has one valence electron.
How many electrons are in a neutral atom?
If the atoms are neutral, they also must contain six electrons. The only difference between these isotopes is the number of neutrons in the nucleus.
What is the neutral lithium atom?
Picture 2.2 Lithium has 3 positve protons in its nucleus. However, a lithium atom is neutral because there are 3 negative electrons outside the nucleus. … Although it is the number of protons that determines the element, the number of electrons will always be the same as the atomic number in a neutral atom.
How many protons does every atom of lithium have quizlet?
Lithium has 3 protons.How many valence electrons are in a neutral lithium atom quizlet?
A lithium atom has 1 valence electron.
How many neutrons are in a atom of MG 25?They differ only because a 24Mg atom has 12 neutrons in its nucleus, a 25Mg atom has 13 neutrons, and a 26Mg has 14 neutrons.
Article first time published onHow many neutrons are in an atom of MG 25 gizmos?
Answer and Explanation: There are 13 neutrons in an atom of magnesium-25. We can determine this by subtracting the number of protons in the atom from the atomic mass which…
How do you find the electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How are the electrons arranged gizmo?
2. Electrons are arranged in orbits called energy levels. The Gizmo shows all of the first two energy levels but only part of the third energy level.
What is neutral atom?
When an atom has an equal number of electrons and protons, it has an equal number of negative electric charges (the electrons) and positive electric charges (the protons). The total electric charge of the atom is therefore zero and the atom is said to be neutral.
How many protons neutrons and electrons are in a neutral lithium atom?
Lithium is an alkali metal with the atomic number = 3 and an atomic mass of 6.941 g/mol. This means that lithium has 3 protons, 3 electrons and 4 neutrons (6.941 – 3 = ~4).
How many stable electrons does lithium have?
Observe: Look at the red lithium atom and the blue oxygen atom. Recall that most atoms are stable when their outermost ring has eight electrons. (Some atoms, such as lithium and beryllium, are stable when their outermost ring has two electrons.)
Why is an atom as a whole neutral?
Electrons have electric charge of -1 and the number of electrons in an atom is equal to the number of protons. … Heavier atoms tend to have more neutrons than protons, but the number of electrons in an atom is always equal to the number of protons. So an atom as a whole is electrically neutral.
How do you know if a atom is neutral?
The amount of charge on a single proton is equal to the amount of charge possessed by a single electron. A proton and an electron have an equal amount but an opposite type of charge. Thus, if an atom contains equal numbers of protons and electrons, the atom is described as being electrically neutral.
How many valence electrons does an atom of Al possess quizlet?
An aluminum atom possesses three valence electrons.
How many valence electrons are in a barium atom?
Barium is an alkaline earth metal; i.e. Group II of the Periodic table. Therefore, it has 2 valence electrons.
How many valence does nitrogen have?
Nitrogen is found to have either 3 or 5 valence electrons and lies at the top of Group 15 on the periodic table. It can have either 3 or 5 valence electrons because it can bond in the outer 2p and 2s orbitals.
How many neutrons does C 14 have?
For example, carbon-14 is a radioactive isotope of carbon that has six protons and eight neutrons in its nucleus. We call it carbon-14 because the total number of protons and neutrons in the nucleus, also known as the mass number, adds up to 14 (6+8=14).
How many neutrons are in the isotope lithium 8?
Unstable known isotopes of lithium include Lithium 8 (5 neutrons), Lithium 9 (6 neutrons), lithium 11 (8 neutrons) and lithium 4 (one neutron).
How many protons are in an atom of Cu quizlet?
What is the atomic number of copper? The element copper has 29 protons.
How many protons neutrons and electrons are in an 25mg 2 ion?
There are 12 protons and 10 electrons in a Mg2+ ion, the normal amount of neutrons is 12 .
How many protons electrons and neutrons are Mg 25?
Magnesium-25 is composed of 12 protons, 13 neutrons, and 12 electrons.
How many protons neutrons and electrons are there in a neutral atom of magnesium 25?
A: Magnesium, in its elemental form, has 12 protons and 12 electrons. The neutrons are a different matter. Magesium’s average atomic mass is 24.305 atomic mass units, but no magnesium atom has exactly this mass.
How many electrons does mg/24 have?
In order to write the Mg electron configuration we first need to know the number of electrons for the Mg atom (there are 12 electrons). When we write the configuration we’ll put all 12 electrons in orbitals around the nucleus of the Magnesium atom.
How many neutrons does mg 22 have?
NuclideZN21Mg12922Mg121023Mg1211
What is the mass number of magnesium 25?
Magnesium-25 atom is the stable isotope of magnesium with relative atomic mass 24.985837, 10.0 atom percent natural abundance and nuclear spin 5/2.
How do you find the number of electrons in a non neutral atom?
To determine how many total electrons there are, add the amount of charge to the atomic number. In this case, there are fewer protons than electrons. For example, N3- has a -3 charge which means it has 3 more electrons than a neutral nitrogen atom. Nitrogen’s atomic number is 7, therefore this ion has 10 electrons.