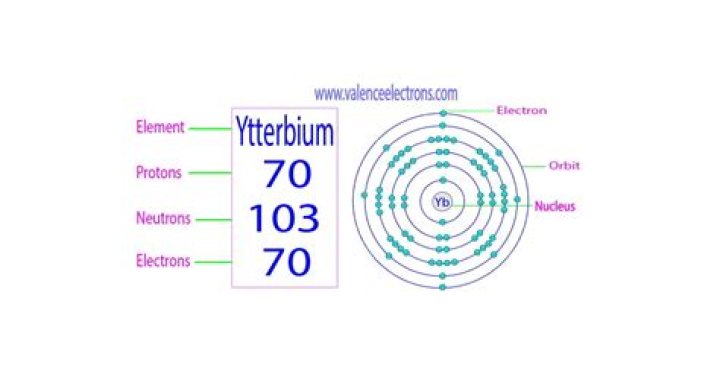
Ytterbium atoms have 70 electrons and the shell structure is 2.8. 18.32. 8.2. The ground state electron configuration of ground state gaseous neutral ytterbium is [Xe].
How many electrons does ytterbium have in total?
Ytterbium atoms have 70 electrons and the shell structure is 2.8. 18.32. 8.2. The ground state electron configuration of ground state gaseous neutral ytterbium is [Xe].
How many valence electrons are in ytterbium?
Most rare-earth metals have a valence of three; however, that of cerium is 3.2, and europium and ytterbium are divalent.
How many neutrons does ytterbium have?
Classification:Ytterbium is a lanthanide and rare earth metalProtons:70Neutrons in most abundant isotope:104Electron shells:2,8,18,32,8,2Electron configuration:[Xe] 4f14 6s2How many electrons are in the 4th shell of ytterbium?
In any case, the elements ytterbium (#70) through oganesson (118)—that is 49 elements of the 118 discovered elements—have 32 electrons in shell 4.
What state of matter is ytterbium?
At room temperature, Ytterbium is in a solid state. As per the elements classification system, Ytterbium is metal. This is a ductile, malleable, and soft element with a silvery luster. It oxidizes when exposed to air and reacts with mineral acids.
What group is Ytterbium in?
GroupLanthanidesMelting pointPeriod6Boiling pointBlockfDensity (g cm−3)Atomic number70Relative atomic massState at 20°CSolidKey isotopes
How is ytterbium 169 made?
Ytterbium 169 is produced by thermal-neutron bombardment of enriched stable ytterbium 168 resulting in a specific activity of greater than 100 mCi/mg. The product contains less than 1 per cent of ytterbium 175 which emits beta and gamma rays and has a 4.2-day half-life.What is the difference between yttrium and ytterbium?
Yttrium is a chemical element having the symbol Y and atomic number 39, while Ytterbium is a chemical element having the symbol Yb and atomic number 70. The key difference between Yttrium and Ytterbium is that natural yttrium is non-radioactive, whereas ytterbium is usually radioactive.
Where is ytterbium found?Ytterbium is found with other rare-earth elements in several rare minerals. It is most often recovered commercially from monazite sand (0.03% ytterbium). The element is also found in euxenite and xenotime. The main mining areas are China, the United States, Brazil, India, Sri Lanka, and Australia.
Article first time published onWhich atom contains exactly 16 neutrons?
if you look at the periodic table, phosphorus has 15 electrons and protons, and 16 neutrons.
What is ytterbium used for?
Ytterbium has few uses. It can be alloyed with stainless steel to improve some of its mechanical properties and used as a doping agent in fiber optic cable where it can be used as an amplifier. One of ytterbium’s isotopes is being considered as a radiation source for portable X-ray machines.
What element family is ytterbium?
Ytterbium belongs to the lanthanide family. The lanthanides make up Row 6 of the periodic table. The periodic table is a chart that shows how the chemical elements are related to each other. The lanthanides are also known as the rare earth elements.
How common is the element ytterbium?
Ytterbium is the 44th most abundant element in the Earth’s crust. It is one of the more common rare earths, present at about 2.7 to 8 parts per million in the crust. It is common in the mineral monazite.
What is the density of the element ytterbium?
Atomic number70Atomic mass173.04 g.mol -1Electronegativity according to Pauling1.1Density7 g.cm-3 at 20°CMelting point824 °C
Does ytterbium oxidize?
ytterbium (Yb), chemical element, a rare-earth metal of the lanthanide series of the periodic table. … It slowly oxidizes in air, forming Yb2O3; the metal is readily dissolved in diluted acids—except hydrofluoric acid (HF), in which a protective layer of YbF3 forms on the surface and impedes further chemical reaction.
What is the hardness of ytterbium?
Melting Point:824°C, 1515.2°F, 1097.15 KPoisson Ratio:(form) 0.207Mohs Hardness:N/AVickers Hardness:206 MPaBrinell Hardness:343 MPa
How many unpaired electrons does ytterbium have?
Number of unpaired electrons in ytterbium is/are. Correct answer is ‘0‘.
What is the 70th element?
Ytterbium is a chemical element with symbol Yb and atomic number 70.
Is ytterbium a noble gas?
The ground state electronic configuration of Neutral Ytterbium atom is [Xe] 4f14 6s2. The portion of Ytterbium configuration that is equivalent to the noble gas of the preceding period, is abbreviated as [Xe].
What does ytterbium react with?
Ytterbium metal reacts with all the halogens to form ytterbium(III) halides. So, it reacts with fluorine, F2, chlorine, Cl2, bromine, I2, and iodine, I2, to form respectively ytterbium(III) bromide, YbF3, ytterbium(III) chloride, YbCl3, ytterbium(III) bromide, YbBr3, and ytterbium(III) iodide, YbI3.
What is the half-life of ytterbium 169?
Ytterbium 169 has a physical half-life of thirty-two days and emits primarily gamma rays (8-308 keV).
What element has the atomic number of 169?
Please visit the Ytterbium element page for information specific to the chemical element of the periodic table.
What is the half-life of gold-198?
Gold-198 decays with a half-life of 2.7 days to stable mercury-198 by emission of ß particles of maximum energy 0.960 Mev and Y -rays of energies 0.412, 0.68 and 1.09 Mev as shown schematically in Figure 1 : In a gold grain, the thickness of the enveloping sheath of platinum is such that the ß particles, but not the Y …