Draw the Lewis structure.Count the total number of bonds.Count the number of bond groups between individual atoms.Divide the number of bonds between atoms by the total number of bond groups in the molecule.
How do you count bonds in molecules?
- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.
How is an S bond formed?
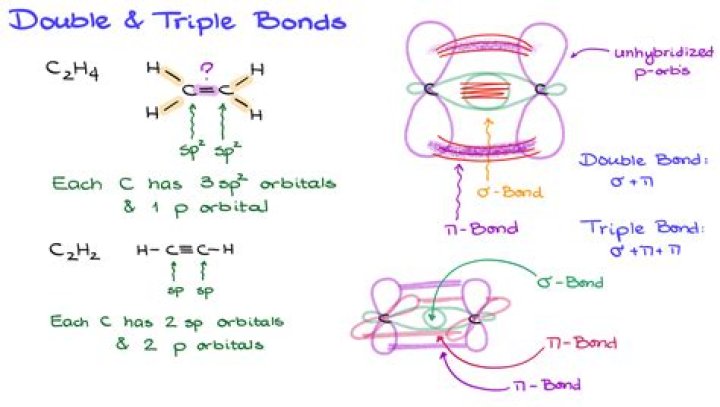
A sigma bond (σ bond) is a bond formed by the overlap of orbitals in an end-to-end fashion, with the electron density concentrated between the nuclei of the bonding atoms.
How many S and P bonds are there in the molecule?
7 sigma and 5 pi.How do you know how many sigma bonds are in a molecule?
Usually, all bonds between atoms in most organic compounds contain one sigma bond each. If it is a single bond, it contains only sigma bond. Double and Triple bonds, however, contains sigma and pi bonds. Double bonds have one each, and triple bonds have one sigma bond and two pi bonds.
What is Bond Order of b2?
Answer: The bond order of B2 molecule is one.
What is the bond order for O2 2?
O2 has two unpaired electrons in its π* orbitals, and a bond order of 2.
How many S and P bonds are there in the molecule of Dicyanoethene?
How many sigma and pi-bonds are there in the molecule of dicyanoethene (CN-CH =CH-CN) ? 3 sigma and 3 pi.How many bonds is a pi bond?
The orientation of the two pi bonds is that they are perpendicular to one another (see Figure 6 below).
How many pi bonds are there in a single bond?The number of pi bonds present in a single bond is zero. Double bond is defined as the chemical bond between two atoms. It forms four bonding electrons between the atoms. The number of sigma bonds present is one and the number of pi bonds present in a double bond is one.
Article first time published onHow many sigma bonds does anthracene have?
There are 24+3-1=26 sigma and 7 pi bonds in Anthracene.
Is Ch a sigma bond?
The C-C sigma bond is formed by the overlap of one sp orbital from each of the carbons, while the two C-H sigma bonds are formed by the overlap of the second sp orbital on each carbon with a 1s orbital on a hydrogen.
What is S and P in chemical bonding?
Sigma and pi bonds are types of covalent bonds that differ in the overlapping of atomic orbitals. … Sigma bonds are a result of the head-to-head overlapping of atomic orbitals whereas pi bonds are formed by the lateral overlap of two atomic orbitals.
How many sigma and pi bonds are in this molecule?
A pi bond is formed by the sideways or lateral overlapping of atomic orbitals. Therefore, they will be the other type of bonds in either a double or triple bond. To answer your question, this molecule has 13 sigma bonds and three pi bonds.
How many sigma bonds are there?
There is no more than 1 sigma bond between any two atoms. Molecules with rings have additional sigma bonds, such as benzene rings, which have 6 C−C sigma bonds within the ring for 6 carbon atoms.
How many sigma bonds are present in p4o10?
The number of sigma bonds (σ-bonds) in P4O10 is 16.
What is the bond order of C2+?
Bond order = 1/2 (number of electrons in bonding orbitals – number of electrons in antibonding orbitals) Therefore, Bond order of C2+ = 1/2 (5 – 2) = 3/2 = 1.5.
What is bond order of NO molecule?
The bond order of NO molecule is 2.
How do you find the number of pi bonds?
Calculation of π-bonds and double bonds (P): where, X = number of carbon atoms; Y = number of hydrogen atoms and P = number of π bonds/double bonds. E.g.: In C176H250, X = 176, Y = 250, therefore P = (2 x 176 – 250)/2 +1 = 51 + 1 = 52 number of π bonds or double bonds.
What are bond angles in chemistry?
A bond angle is the angle between any two bonds that include a common atom, usually measured in degrees. A bond distance (or bond length) is the distance between the nuclei of two bonded atoms along the straight line joining the nuclei.
How many pi bonds are present in CH3CN?
There are a total of 5 sigma bonds and 2 pi bonds in CH3CN.
How many sigma and pi bonds are there in cyanogenmod?
Cyanogen, N≡C−C≡N N ≡ C − C ≡ N has three sigma and four ‘pi’ bonds.
How many bonds does C2 have?
The bonding components in C2 consist of two weakly bonding σ bonds and two electron-sharing π bonds.
Do lone pairs count as pi bonds?
However that lone pair is not part of the pi bond.
Can s orbitals form pi bonds?
A π bond has a plane of symmetry along the bond axis. It cannot be formed by s-orbitals; it needs at least p-orbitals to be created.
How many σ bonds and π bonds are there in a anthracene molecule?
The chemical formula of anthracene is C14H10 C 14 H 10 . It can be seen that 7 double bonds (pi-bond) and 26…
How many pi electrons does anthracene have?
4.1.2. Anthracene contains 7 C=C bonds in the closed loop Anthracene contain 14 π electrons.
How many pi bonds are in DNB?
It has five π-bonds.
How many bond is present in H CC CH CH ch3?
6σ,6π
How many sigma bonds are in ch3?
8σ,3π
How many pi bonds are there in ethene?
∴ Ethene molecule has 1 pi and 5 sigma bonds.