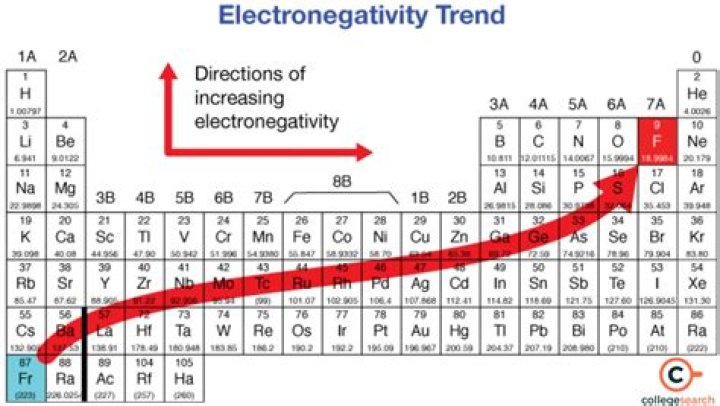
Elements from the halogen group including F, Cl, Br have pretty high electronegativities. The most electronegative element is Fluorine with a score of 4.0 (the highest possible.) Across from Fluorine we also have N and O with high electronegativities.
What are the 3 most electronegative elements?
And those three elements are fluorine, oxygen, and nitrogen. And in fact, the reason why they’re capable is hydrogen bonds is because these three are the most electronegative elements. So again, this is a mnemonic you can use to memorize the most electronegative elements on the periodic table.
What is the most electronegative element?
Fluorine is assinged the oxidation number of -1 because it attracts the electrons in the bond more strongly than the carbon does. Thus, fluorine appears to have an extra electron, -1 oxidation number. F is the most electronegative element on the periodic table.
What element has the highest electronegativity in group 3?
In Period 3, sodium with 11 protons is the least electronegative element, and chlorine with 17 protons is the most electronegative element. You might expect argon (with 18 electrons) to be the most electronegative element in Period 3.Where are the highest electronegativity elements found?
On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. As a result, the most electronegative elements are found on the top right of the periodic table, while the least electronegative elements are found on the bottom left.
Which element has the highest electronegativity in the metalloids?
The elements classified as metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium. Which of the following elements is most electronegative? Chlorine has the greatest electronegativity because out of all the choices, it lies farthest to the right and top of periodic table.
What is K electronegativity?
The first scale of electronegativity was developed by Linus Pauling and on his scale potassium has a value of 0.82 on a scale running from from about 0.7 (an estimate for francium) to 2.20 (for hydrogen) to 3.98 (fluorine).
What is electronegativity chemistry?
Electronegativity is a chemical property that describes the tendency of an atom or a functional group to attract electrons toward itself.What is the electronegativity of all elements?
NUMBERSYMBOLELECTRONEGATIVITY1H2.202Heno data3Li0.984Be1.57
Do metals have high electronegativity?Since metals have few valence electrons, they tend to increase their stability by losing electrons to become cations. Consequently, the electronegativities of metals are generally low.
Article first time published onWhat element is the largest?
Thus, helium is the smallest element, and francium is the largest.
Is a higher number more electronegative?
An atom’s electronegativity is affected by both the element’s atomic number and its size. The higher its electronegativity, the more an element attracts electrons.
What are the elements located at the highest points?
The chemical element with the lowest melting point is Helium and the element with the highest melting point is Carbon.
Which elements have the highest electronegativity metals or nonmetals?
Nonmetals have much higher electronegativities than metals; of the nonmetals, fluorine is the most electronegative, followed by oxygen, nitrogen, and chlorine. The larger the difference in electronegativity between two atoms, the more polar the bond between them.
Which has the highest electronegativity fluorine or chlorine?
Fluorine is more electronegative than chlorine because fluorine is smaller and has its electrons closer to the positively charged nucleus. …
Which has the highest electronegativity SR or RA?
H 2.1K 0.9Ca 1.0V 1.5Rb 0.9Sr 1.0Nb 1.5Cs 0.8Ba 1.0Ta 1.4Fr 0.8Ra 1.0
Which element has the highest electronegativity quizlet?
Fluorine (F) has the highest electronegativity (4.0). It does take electrons from the atoms of other elements.
Which has highest electron affinity?
Which Element Has the Highest Electron Affinity? Chlorine has the highest electron affinity among the elements. Its high affinity can be attributed to its large atomic radius, or size. Because chlorine’s outermost orbital is 3p, its electrons have a large amount of space to share with an incoming electron.
What has the highest ionization energy?
Thus, helium has the largest first ionization energy, while francium has one of the lowest.
Which list of elements is written in order of increasing electronegativity?
- Electronegativity increases left to right across a row in the periodic table e.g. C < N < O < F.
- Electronegativity decreases as you move down a group in the periodic table e.g. F > Cl > Br > I.
- F is the most electronegative element.
Why does electronegativity increase across Period 3?
Electronegativity increases across a period because the number of charges on the nucleus increases. That attracts the bonding pair of electrons more strongly.
Which functional group is the most electronegative?
The carbonyl group (“carbon double bond oxygen”) is polar since oxygen is more electronegative than carbon and forms a partially charged dipole.
Which element has the highest electronegativity and why?
Electronegativity increases from bottom to top in groups, and increases from left to right across periods. Thus, fluorine is the most electronegative element, while francium is one of the least electronegative.
Why do some elements have a higher electronegativity?
An atom’s electronegativity is affected by both its atomic number and the size of the atom. The higher its electronegativity, the more an element attracts electrons. … The nuclear charge is important because the more protons an atom has, the more “pull” it will have on negative electrons.
Why do nonmetals have higher electronegativity?
Non-Metals are generally more electronegative due to their Smaller atomic radii. Non-metals having high pull forces on their electron because they are closer to having a full valance shell, which makes them stable. The much number of valance electrons have more electronegative.
Which is the biggest element and why?
The largest element of the periodic table is Francium. The alkali metals are present on the extreme left of the periodic table and have the largest size in a given period.
What is the largest element in period 3?
It is found that Helium is the smallest element and Francium is the largest element.
What element has the smallest electronegativity?
The element with the lowest electronegativity value is francium, which has an electronegativity of 0.7. This value uses the Pauling scale to measure electronegativity.
Which elements are more electronegative than carbon?
Bromine is more electronegative than carbon and so the bond is polarised in the way that we have already described with C-F and C-Cl. The polarity of the carbon-halogen bonds is important in the reactions of the halogenoalkanes.
Which element in each of the following pairs is more electronegative?
Br is more electronegative than Be. In general, electronegativity increases from left to right in the periodic table, so elements in group 17 (such as Br) are more electronegative than elements in group 2 (such as Be).
How do you find the electronegativity difference between three elements?
Subtract the smaller electronegativity from the larger one to find the difference. For example, if we’re looking at the molecule HF, we would subtract the electronegativity of hydrogen (2.1) from fluorine (4.0). 4.0 – 2.1 = 1.9. If the difference is below about 0.5, the bond is nonpolar covalent.