Crystalloids have small molecules, are cheap, easy to use, and provide immediate fluid resuscitation
What are examples of Crystalloids?
The most frequently used crystalloid fluid is sodium chloride 0.9%, more commonly known as normal saline 0.9%. Other crystalloid solutions are compound sodium lactate solutions (Ringer’s lactate solution, Hartmann’s solution) and glucose solutions (see ‘Preparations containing glucose’ below).
What are colloid IV fluids?
Colloids and crystalloids are types of fluids that are used for fluid replacement, often intravenously (via a tube straight into the blood). Crystalloids are low-cost salt solutions (e.g. saline) with small molecules, which can move around easily when injected into the body.
What are Crystalloids?
A crystalloid fluid is an aqueous solution of mineral salts and other small, water-soluble molecules. Most commercially available crystalloid solutions are isotonic to human plasma. These fluids approximate concentrations of various solutes found in plasma and do not exert an osmotic effect in vivo.Are colloids and Crystalloids the same?
Colloids are those substances which are not easily crystallized from their aqueous solutions. Crystalloids are those substances which are easily crystallized from their aqueous solution. Colloids contain much larger particles than crystalloids (1 – 200 nm).
What does colloidal mean?
1 : a gelatinous or mucinous substance found in tissues in disease or normally (as in the thyroid) 2a : a substance consisting of particles that are dispersed throughout another substance and are too small for resolution with an ordinary light microscope but are incapable of passing through a semipermeable membrane.
What are the 4 types of colloids?
- Sol is a colloidal suspension with solid particles in a liquid.
- Emulsion is between two liquids.
- Foam is formed when many gas particles are trapped in a liquid or solid.
- Aerosol contains small particles of liquid or solid dispersed in a gas.
What are the three types of Crystalloids?
Types of Crystalloid Solutions There are three tonic states: isotonic, hypertonic, and hypotonic.Is dextrose a crystalloid?
Crystalloids are the most common fluids used in the healthcare setting. The following are some examples of the most common solutions in the crystalloid category. Dextrose 5% is consists of 278 mmoL/L of dextrose. The pH is 4.0 and the osmolarity is around 272.
What are the 3 main types of IV fluids?- Isotonic Solutions. Isotonic solutions are IV fluids that have a similar concentration of dissolved particles as blood. …
- Hypotonic Solutions. Hypotonic solutions have a lower concentration of dissolved solutes than blood. …
- Hypertonic Solutions.
What are the advantages of Crystalloids?
The advantage of crystalloid fluid resuscitation is that volume has not only been lost from the intravascular space, but also extracellular water has been drawn to the intravascular space by oncotic pressure. Solutions with lower sodium concentrations distribute more evenly throughout the total body water.
What is difference between crystal and crystalloid?
As nouns the difference between crystalloid and crystal is that crystalloid is any substance that can be crystallized from solution while crystal is (countable) a solid composed of an array of atoms or molecules possessing long-range order and arranged in a pattern which is periodic in three dimensions.
Is FFP a colloid?
Colloids have larger molecules and may be more efficient at increasing fluid volume in the blood. They include starches, dextrans, gelatins, and naturally-occurring colloids, such as albumin or fresh frozen plasma or FFP.
What systems are called colloidal?
A colloidal system contains two separate phases: a dispersed phase (or internal phase) and a continuous phase (or dispersion medium). The part which is dispersed is known as the dispersed phase and is suspended in the continuous phase. … Food colloids are sols, gels, emulsion, and foam.
What are 5 examples of colloid?
Colloids are common in everyday life. Some examples include whipped cream, mayonnaise, milk, butter, gelatin, jelly, muddy water, plaster, colored glass, and paper. Every colloid consists of two parts: colloidal particles and the dispersing medium.
What are 10 examples of colloids?
Class of ColloidDispersed PhaseExamplesSolid aerosolsolidsmoke, dust in airSolid emulsionliquidcheese, butterLiquid emulsionliquidmilk, mayonnaiseLiquid aerosolliquidfog, mist, clouds, aerosol spray
What are the 7 types of colloids?
- Foam.
- Solid Foam.
- Aerosol.
- Emulsion.
- Gel.
- Solid Aerosol.
- Sols.
- Solid sols.
What is optic colloid?
Optical Properties of Colloids When a beam of light is passed through a colloidal solution kept in dark, the path of the beam gets illuminated with blue colour. … The Tyndall effect is due to the scattering of light by colloidal particles. Tyndall effect is not exhibited by a true solution.
What is the best describe of colloids?
Colloids are mixtures in which one or more substances are dispersed as relatively large solid particles or liquid droplets throughout a solid, liquid, or gaseous medium. The particles of a colloid remain dispersed and do not settle due to gravity, and they are often electrically charged.
Is glucose a crystalloid?
Crystalloid intravenous fluids, which include solutions containing small molecular weight solutes such as sodium, chloride and glucose, are the most common type of fluid used to replace blood in the United States.
What is d5 saline?
Dextrose and Sodium Chloride Injection, USP (dextrose and sodium chloride inj) is a sterile, nonpyrogenic solution for fluid and electrolyte replenishment and caloric supply in single dose containers for intravenous administration. It contains no antimicrobial agents.
Why are Crystalloids used in sepsis?
Answer: Crystalloid solutions remain the resuscitative fluid of choice for patients with sepsis and septic shock. Balanced crystalloid solutions may improve patient-centered outcomes and should be considered as an alternative to 0.9% normal saline (when available) in patients with sepsis.
Is lactated Ringer's crystalloid?
Ringer’s lactate solution, or lactated Ringer’s solution, is a type of isotonic, crystalloid fluid further classified as a balanced or buffered solution used for fluid replacement.
What is the difference between crystalloid and colloid fluids?
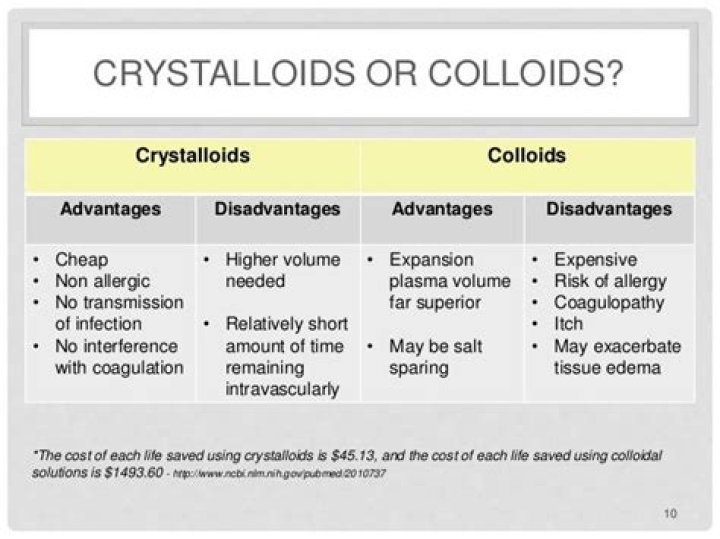
Crystalloids have small molecules, are cheap, easy to use, and provide immediate fluid resuscitation, but may increase oedema. Colloids have larger molecules, cost more, and may provide swifter volume expansion in the intravascular space, but may induce allergic reactions, blood clotting disorders, and kidney failure.
What are lactated ringers?
The contents of Ringer’s lactate include sodium, chloride, potassium, calcium, and lactate in the form of sodium lactate, mixed into a solution with an osmolarity of 273 mOsm/L and pH of about 6.5. In comparison, normal saline (NS) has an osmolarity of about 286 mOsm/L.
What are the types of IV?
- Normal Saline.
- Half Normal Saline.
- Lactated Ringers.
- Dextrose.
How long does LR stay intravascular?
In contrast, the T1/2 during the postoperative period is usually short, about 15 to 20 min, at least in response to new fluid. The commonly used colloid fluids have an intravascular persistence T1/2 of 2 to 3 h, which is shortened by inflammation.
Is D5W a crystalloid or colloid?
Dextrose 5% in Water (D5 or D5W, an intravenous sugar solution) A crystalloid that is both isotonic and hypotonic, administered for hypernatremia and to provide free water for the kidneys. Initially hypotonic, D5 dilutes the osmolarity of the extracellular fluid.
Which of the following colloids are called protective colloids?
Lyophilic colloids have a unique property of protecting lyophobic colloids. When a lyophilic sol is added to the lyophobic sol, the lyophilic particles form a layer around lyophobic particles and thus protect the latter from electrolytes. Lyophilic colloids use for this purpose are called protective colloids.
Which is natural colloid?
Which is a natural colloid ? Explanation : Blood is a natural colloid. Sodium chloride solution, cane sugar solution and urea solution are artificially prepared.
Is LR a colloid?
Crystalloid solutions are the most frequently chosen, by far, with normal saline (NS) and lactated Ringer’s (LR) both being the most frequent choices. Colloids are an alternative to crystalloids, with highly variable use depending on a myriad of clinical variables.