It is the blocking of COX-1 that is responsible for the increased risk of stomach irritation when aspirin is taken, as it stops the formation of a protective prostaglandin (prostacyclin), in cells in the stomach lining, which reduces gastric acid production.
Which functional groups Does an aspirin molecule contain?
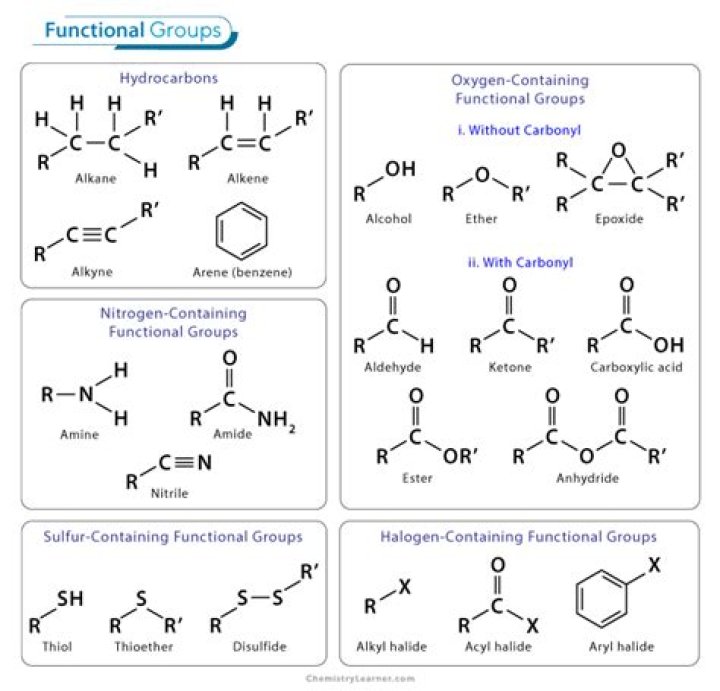
There are three different functional groups in aspirin, which contribute to its properties, a weak acid soluble in basic solutions. These functional groups include carboxylic acid, ester, and an aromatic group.
What two functional groups are contained in an aspirin molecule quizlet?
The functional groups that are different between salicylic acid and aspirin: Salicylic acid has an OH group, and aspirin has an ester.
What is the ester group in aspirin?
Aspirin is both an aromatic carboxylic acid (red oval) and a phenyl ester of acetic acid (blue oval). While esterification will convert the carboxylic acid group to a methyl ester, transesterification (exchange of one alcohol portion of an ester for another alcohol) to afford methyl acetate 4 and methyl salicylate 3.Why is aspirin less irritating than salicylic acid?
Salicylic acid contains a phenol group, and phenols are known to be irritating. The Bayer Company replaced the phenol group with an ester group. This esterified compound (acetylsalicylic acid, also known as aspirin) was shown to be much less irritating than salicylic acid.
Does aspirin have a ketone group?
The male sex hormone testosterone contains ketone, alkene, and secondary alcohol groups, while acetylsalicylic acid (aspirin) contains aromatic, carboxylic acid, and ester groups.
Does aspirin have an ether functional group?
In the case of aspirin, the ether group is a combination of a simple phenol group (OH) and acetic acid (C2H4O2). When the ether group in aspirin is hydrolyzed, acetic acid is formed, and the aspirin molecule becomes salicylic acid.
What is aromatic functional group?
Aromatics. The additional functional group that contains only carbon and hydrogen is an aromatic ring which is a six-carbon ring with alternative double bonds. The aromatic ring is also shown as a ring with a circle in the middle representing the double bonds. … 2: Aromatic rings contain alternating double bonds.What is phenol functional group?
Functional group in Phenol is a hydroxyl group (-OH) attached to a benzene ring to a sp2 hybridized carbon of benzend ring..
Which functional group is found in an ester?ester: A compound most often formed by the condensation of an alcohol and an acid, with elimination of water. It contains the functional group C=O. joined via carbon to another oxygen atom. ether: Compound containing an oxygen atom bonded to two hydrocarbon groups.
Article first time published onWhat 2 functional groups are on the salicylic acid?
Salicylic acid (2-hydroxybenzoic acid) is formed of a benzene ring to which 2 adjacent groups, carboxylic group and hydroxy group, are attached. We don’t normally consider benzene to be a functional group, so that’s why the hydroxyl and carboxylic are the ones that count.
What quantity of aspirin is contained in most over the counter aspirin products?
A standard tablet contains 5 grains of aspirin, which is equal to 325 milligrams of the drug. Aspirin advertised as “arthritis strength” or “extra strength” contains more aspirin per tablet (usually 500 mg).
Which general formula represents the functional group in a carboxylic acid quizlet?
an organic compound that contains a carboxyl group (C(O)OH). [1] The general formula of a carboxylic acid is R−C(O)OH with R referring to the rest of the (possibly quite large) molecule.
Is aspirin absorbed in the stomach or intestine?
Aspirin was not absorbed by the stomach at pH 6.5. These experiments indicate that aspirin can be absorbed to an appreciable extent in its ionized form by the small intestine but not by the stomach.
Why is aspirin bad for your stomach chemistry?
However, aspirin also inhibits COX-1 activity in the gastric mucosa by suppressing the synthesis of protective prostaglandins. In doing so, this creates a state of propensity for the development of aspirin-associated gastrointestinal ulcers and ulcer complications.
Does aspirin have a phenol group?
No, because the aspirin does not have a phenol group. The ferric chloride gives a violet color with any molecule possessing a phenol group.
Does aspirin have an ester?
Aspirin is an ester which passes through the stomach unchanged before being hydrolysed by the basic medium of the intestine to form the active compound. When a carboxylic acid reacts with an alcohol (such as ethanol or a phenol), the products are water and an ester.
What is the primary function of aspirin?
Aspirin is a common drug for relieving minor aches, pains, and fevers. People also use it as an anti-inflammatory or a blood thinner. People can buy aspirin over the counter without a prescription. Everyday uses include relieving headache, reducing swelling, and reducing a fever.
Is aspirin soluble in hexane?
Acetylsalicylic acid is fairly polar and not soluble in pure hexane. Ethyl acetate (CH3CO2CH2CH3) is much more polar and your compound is very soluble in pure ethyl acetate.
Does aspirin have an alcohol functional group?
acetylsalicylic acid, is now known as aspirin and is shown below (left), next to the structure for salicylic acid (center). Note that the salicylic acid has an organic acid functional group, and an alcohol group, on an aromatic hydrocarbon ring.
What does aspirin react with?
Some products that may interact with this drug include: mifepristone, acetazolamide, “blood thinners” (such as warfarin, heparin), corticosteroids (such as prednisone), dichlorphenamide, methotrexate, valproic acid, herbal medications (such as ginkgo biloba).
What is the functional group of resorcinol?
Resorcinol is a benzenediol that is benzene dihydroxylated at positions 1 and 3. It has a role as an erythropoietin inhibitor and a sensitiser. It is a benzenediol and a member of resorcinols. Resorcinol is a 1,3-isomer (or meta-isomer) of benzenediol with the formula C6H4(OH)2.
What is thiol functional group?
Thiol (mercaptan): A functional group characterized by a sulfhydryl group bonded to a carbon of any hybridization that is not a carbonyl group carbon.
Which functional group is present in resorcinol?
Hint: The structure of resorcinol includes two hydroxyl groups on the benzene ring. The two hydroxyl groups are separated by one carbon atom.
Are alkyl groups functional groups?
An alkyl is a functional group of an organic chemical that contains only carbon and hydrogen atoms, which are arranged in a chain.
What is benzaldehyde functional group?
Benzaldehyde (C6H5CHO) is an organic compound consisting of a benzene ring with a formyl substituent. It is the simplest aromatic aldehyde and one of the most industrially useful.
Is carbonyl a functional group?
A carbonyl group is a chemically organic functional group composed of a carbon atom double-bonded to an oxygen atom –> [C=O] The simplest carbonyl groups are aldehydes and ketones usually attached to another carbon compound.
Which contains the carbonyl group?
Carbon directly attached to oxygen with a double bond is said to be carbonyl group. As aldehyde, ketones, carboxylic acids, ester, acid halides, acid anhydrides all comprise of a carbonyl group therefore the correct option is D.
Is methyl a functional group?
The methyl group is the only nonpolar functional group in our class list above. The methyl group consists of a carbon atom bound to three hydrogen atoms. … This means that methyl groups are unable to form hydrogen bonds and will not interact with polar compounds such as water.
Is ethyl a functional group?
Chemical classEsterGroupEsterFormulaRCO2R’Prefix/ExampleEthyl acetate (Ethyl ethanoate)
Which functional group or groups are present in both salicylic acid and aspirin?
The structures look quite similar. They both have a benzene ring carrying two groups, on adjacent carbon atoms. In both of them one of the groups is a carboxylic acid group. But, salicylic acid carries a phenol group whilst aspirin does not.