In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. There are 18 numbered groups in the periodic table; the f-block columns (between groups 2 and 3) are not numbered. … For example, group 16 is also described as the “oxygen group” and as the “chalcogens”.
What is a family of elements?
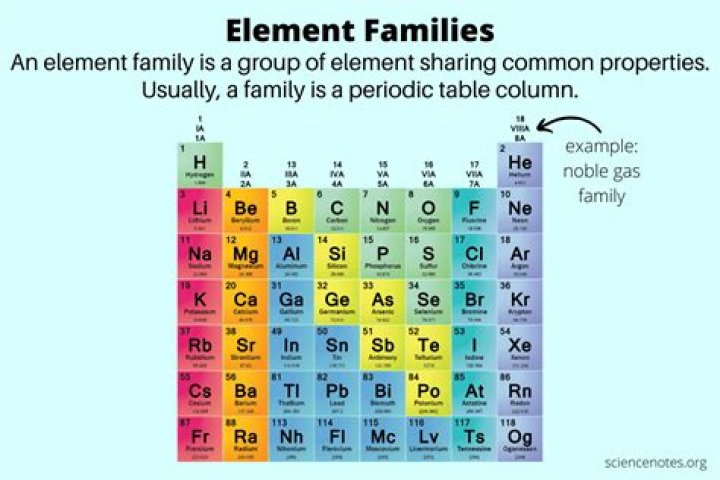
An element family is a column of elements on the periodic table. Each member of a family has the same number of valence electrons. Family members share similar chemical and physical properties. An element family is also called an element group.
What are the group of elements?
- Group 1: Hydrogen and the Alkali Metals.
- Group 2: The Alkaline Earth Metals.
- Group 3: Transition Metals.
- Group 4: Transition Metals.
- Group 5: Transition Metals.
- Group 6: Transition Metals.
- Group 7: Transition Metals.
- Group 8: Transition Metals.
What are the names of each family of elements?
- Group 1: alkali metals, or lithium family.
- Group 2: alkaline earth metals, or beryllium family.
- Group 3: the scandium family.
- Group 4: the titanium family.
- Group 5: the vanadium family.
- Group 6: the chromium family.
- Group 7: the manganese family.
- Group 8: the iron family.
What are the three families of elements?
Recall that all elements can be classified into three categories: metals, nonmetals, and semiconductors. Scientists classify the elements further into five families. The atoms of all elements in most families have the same number of valence electrons.
How many families of elements are there in the periodic table?
5 Element Families The five element families combine similar element groups. So, even though there are numerous columns of transition metals on the periodic table, they all belong to the same family. The transition metals also include the lanthanides and actinides, which appear the main body of the table.
Are groups and families the same thing?
For the most part, element families and element groups are the same things. Both describe elements that share common properties, usually based on the number of valence electrons. Usually, either family or group refers to one or more columns of the periodic table.
What is the name of the family for elements in Group 17?
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).Which elements are in the same family?
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties.
What are group 7 elements called?Group 7A (or VIIA) of the periodic table are the halogens: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The name “halogen” means “salt former”, derived from the Greek words halo- (“salt”) and -gen (“formation”).
Article first time published onWhat is another name for groups of elements?
A group is a column of elements in the periodic table of the chemical elements. A group is also called a family.
What are the representative elements usually called?
In chemistry and atomic physics, the main group is the group of elements (sometimes called the representative elements) whose lightest members are represented by helium, lithium, beryllium, boron, carbon, nitrogen, oxygen, and fluorine as arranged in the periodic table of the elements.
What is a representative element?
The representative elements are elements where the s and p orbitals are filling. The transition elements are elements where the d orbitals (groups 3–11 on the periodic table) are filling, and the inner transition metals are the elements where the f orbitals are filling.
What is the name of the family of the elements in group 16?
The oxygen family, also called the chalcogens, consists of the elements found in Group 16 of the periodic table and is considered among the main group elements. It consists of the elements oxygen, sulfur, selenium, tellurium and polonium. These can be found in nature in both free and combined states.
What is the name of the family of elements in Group IA 1?
Group 1A — The Alkali Metals. Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).
What is not an element family group?
Hydrogen is a very special element of the periodic table and doesn’t belong to any family. While hydrogen sits in Group I, it is NOT an alkali metal.
Is a family a group?
A family is a group of people related by the ties of blood, marriage, or adoption. Members of a family most often live in a single residence and perform different duties. Members of a family and their duties vary from culture to culture and at different times in history.
What family of elements does arsenic as belong?
arsenic (As), a chemical element in the nitrogen group (Group 15 [Va] of the periodic table), existing in both gray and yellow crystalline forms.
What are the Group 6 elements called?
Group 6A (or VIA) of the periodic table are the chalcogens: the nonmetals oxygen (O), sulfur (S), and selenium (Se), the metalloid tellurium (Te), and the metal polonium (Po).
What are the different families on the periodic table and what are their characteristics?
- The IA family is made up of the alkali metals. In reactions, these elements all tend to lose a single electron. …
- The IIA family is made up of the alkaline earth metals. …
- The VIIA family is made up of the halogens. …
- The VIIIA family is made up of the noble gases.
What are the 10 families of elements?
Related elements, including the noble gases, halogens, alkali metals, alkaline earth metals, transition metals, lanthanides, and actinides. In addition, metals, nonmetals, and metalloids form loosely defined families. Other family designations—such as carbon family—are sometimes used.
What does family mean in chemistry?
Chemical Family means a group of elements in the Periodic Table or, more commonly, compounds that share certain physical and chemical characteristics and have a common name.
What is the family of salt producing elements?
Being in the first column, sodium is a member of the alkali metal family with potassium (K) and lithium (Li). Sodium’s big claim to fame is that it’s one of two elements in your table salt. When bonded to chlorine (Cl), the two elements make sodium chloride (NaCl).
What is the family name of group 18?
The noble gases (Group 18) are located in the far right of the periodic table and were previously referred to as the “inert gases” due to the fact that their filled valence shells (octets) make them extremely nonreactive.
What is Group 18 called?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
What is the name of the family in group 2?
Alkaline earth metals – The metals of group 2: Be, Mg, Ca, Sr, Ba, Ra.
What are the group 8 elements called?
Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn).
What are the Group 4 elements called?
Group 4 is the second group of transition metals in the periodic table. It contains the four elements titanium (Ti), zirconium (Zr), hafnium (Hf), and rutherfordium (Rf). The group is also called the titanium group or titanium family after its lightest member.
What are the group 5 elements called?
IUPAC group number5Name by elementvanadium groupCAS group number (US, pattern A-B-A)VBold IUPAC number (Europe, pattern A-B)VA
How many groups Family and period does a periodic table have?
Groups are the columns of the periodic table, and periods are the rows. There are 18 groups, and there are 7 periods plus the lanthanides and actinides.
Which is another name for groups of elements actinides?
At the bottom of the periodic table is a special group of metallic radioactive elements called actinides or actinoids. These elements, usually considered ranging from atomic number 89 to atomic number 103 on the periodic table, have interesting properties, and play a key role in nuclear chemistry.