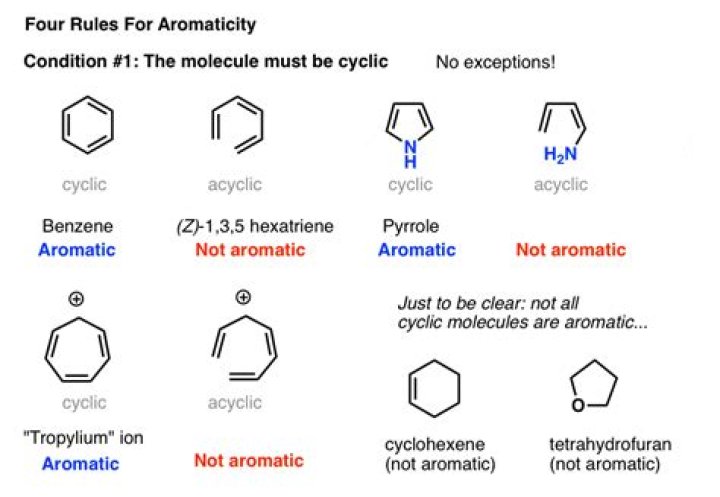
An aromatic molecule or compound is one that has special stability and properties due to a closed loop of electrons. Not all molecules with ring (loop) structures are aromatic. … Aromatic molecules are sometimes referred to simply as aromatics.
How is aromaticity defined?
In chemistry, aromaticity is a property of cyclic (ring-shaped), typically planar (flat) structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to other geometric or connective arrangements with the same set of atoms.
What are aromatic compounds with example?
Aromatic compounds are chemical compounds that consist of conjugated planar ring systems accompanied by delocalized pi-electron clouds in place of individual alternating double and single bonds. They are also called aromatics or arenes. The best examples are toluene and benzene.
Why is it called aromatic?
Aromatic compounds, originally named because of their fragrant properties, are unsaturated hydrocarbon ring structures that exhibit special properties, including unusual stability, due to their aromaticity. They are often represented as resonance structures containing single and double bonds.How do you know if a compound is aromatic?
Explanation: For a compound to be considered aromatic, it must be flat, cyclic, and conjugated and it must obey Huckel’s rule. Huckel’s rule states that an aromatic compound must have pi electrons in the overlapping p orbitals in order to be aromatic (n in this formula represents any integer).
What is the word for aromatic?
Some common synonyms of aromatic are fragrant, odorous, and redolent. While all these words mean “emitting and diffusing scent,” aromatic applies to things emitting pungent often fresh odors.
Why is pyrrole aromatic?
Pyrrole is cyclic and conjugated (that lone pair on nitrogen can contribute to the pi-system). There are two pi bonds and one lone pair of electrons that contribute to the pi system. This gives us 6 total pi electrons, which is a Huckel number (i.e. satisfies 4n+2). Therefore it’s aromatic.
What are aromatic properties?
Aromaticity is a chemical property in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibit a stabilization stronger than would be expected by the stabilization of conjugation alone. It can also be considered a manifestation of cyclic delocalization and of resonance.What are aromatic plants?
Aromatic plants are a special kind of plants used for their aroma and flavour. Many of them are also used for medicinal purposes. Aromatic plants are from a numerically large group of economically important plants. … Aromatic compounds are present in plants i.e. in root, wood, bark, foliage, flower, fruit, seed etc.
Is Naphthalene a liquid?Naphthalene (NAF-thuh-leen) is a white crystalline volatile solid with a characteristic odor often associated with mothballs. The compound sublimes (turns from a solid to a gas) slowly at room temperature, producing a vapor that is highly combustible.
Article first time published onWhat are aromatic compounds easy definition?
Aromatic compounds are substances that consist of one or more rings that contain alternating single and double bonds in its chemical structure. … For example, benzene is an aromatic compound. The chemical structure of benzene, as illustrated here, contains a hexagon ring with alternating double bonds.
How many compounds are aromatic?
Hence the total number of aromatic compounds is 4. Molecules which are cyclic, planar, and have 4nπ electrons which are in conjugation are called anti-aromatic. The compounds which do not follow huckel’s rule for aromaticity and anti-aromaticity are non-aromatic. Aromatic compounds generally have distinctive aroma.
What is aromatic and non aromatic compounds?
A molecule is aromatic if it is cyclic, planar, completely conjugated compound with 4n + 2 π electrons. It is antiaromatic if all of this is correct except it has 4n electrons, Any deviation from these criteria makes it non-aromatic.
Why Benzene is an aromatic compound?
Benzene is an aromatic hydrocarbon because it obeys Hückel’s rule. … It is now considered aromatic because it obeys Hückel’s rule: 4n+2 = number of π electrons in the hydrocarbon, where n must be an integer. In the case of benzene, we have 3 π bonds (6 electrons), so 4n+2=6 .
What is aromaticity and Huckel rule?
In 1931, German chemist and physicist Erich Hückel proposed a theory to help determine if a planar ring molecule would have aromatic properties. His rule states that if a cyclic, planar molecule has 4n+2 π electrons, it is considered aromatic. This rule would come to be known as Hückel’s Rule.
Why is naphthalene aromatic?
As we are getting a whole number from, 4nπ+2 formula for naphthalene, so naphthalene is aromatic. Therefore, the naphthalene is an aromatic compound according to Huckel’s rule because it has 4nπ+2 delocalised electrons. Note: Pi bonds are known as delocalized bonds. Pi bonds cause the resonance.
Are purines aromatic?
Purine is a heterocyclic aromatic organic compound that consists of two rings (pyrimidine and imidazole) fused together. … They are the most widely occurring nitrogen-containing heterocycles in nature.
Is naphthalene an aromatic compound?
Naphthalene, with two fused rings, is the simplest polycyclic aromatic molecule. Note that all the carbon atoms except those at the points of fusion have a bond to a hydrogen atom. Naphthalene, which has 10 π electrons, satisfies the Hückel rule for aromaticity.
Which is aromatic alcohol?
The aromatic alcohols are those compounds in which the hydroxyl group is not directly attached to the nucleus but is linked to a carbon atom situated in a side-chain. … The oxidation products of the aromatic primary alcohols are aldehydes or carboxylic acids and those of aromatic secondary alcohols are ketones.
What is the base word for aromatic?
Aromatic derives from the Greek aromatikos, meaning “sweet spice.” It is related to aroma, but there is one key difference between the meanings of the two words.
What does aromatic smell like?
By definition, aromatic perfumes are a blend of cumin, lavender, sage, rosemary and many unique plant varieties with a strong scent of herbs and spices. Most of the time you will come across aromatic scents that have intense spicy and grassy smells. These fragrances are high in spicy and citrusy notes.
Why are plants aromatic?
Aromatic plants are those that have volatile oils in their leaves and stems. When brushed against or their foliage is bent or crushed, the scents are released. These types of plants are often cultivated for their essential oils.
What is aromatic and medicinal plants?
Medicinal & Aromatic Plants (MAPs) are botanical raw materials, also known as herbal drugs, that are primarily used for therapeutic, aromatic and/or culinary purposes as components of cosmetics, medicinal products, health foods and other natural health products.
What are aromatic leaves?
Aromatics are foliage plants that contain high oil content. During dry times between rainfall seasons these plants utilize their internal oils to keep leaves from withering. Many of our Mediterranean herbs such as lavender are aromatic, and growing them means you have a lot of cutting material to work with.
What are aromatic chemicals used for?
Aromatics are used to make products for areas as diverse as medicine, hygiene, transport, telecommunications, fashion and sports.
What are the main sources of aromatic hydrocarbons?
Simple aromatic hydrocarbons come from two main sources: Coal and petroleum. Coal is a complex mixture of a large number of compounds, most of which are long-chain compounds. If coal is heated to about 1000 °C in the absence of air (oxygen), volatile components, the so-called tar oil, are stripped out.
Is naphthalene an atom?
naphthalene, the simplest of the fused or condensed ring hydrocarbon compounds composed of two benzene rings sharing two adjacent carbon atoms; chemical formula, C10H8.
Is naphthalene a molecule or ion?
A naphthalene molecule can be viewed as the fusion of a pair of benzene rings. (In organic chemistry, rings are fused if they share two or more atoms.) As such, naphthalene is classified as a benzenoid polycyclic aromatic hydrocarbon (PAH).
Is naphthalene molecular or ionic?
Naphthalene is a molecular solid. If the crystals contains only individuals atoms; as in solid argon or krypton or if they are composed of non polar molecules as in naphthalene, the only attraction between the molecules are the London forces. Name an important source from which naphthalene balls are obtained.
What are aromatic amino groups?
An aromatic amino acid is an amino acid that includes an aromatic ring. Among the 20 standard amino acids, the following are classically considered aromatic: phenylalanine, tryptophan and tyrosine.
What is not aromatic?
Cyclooctatetraene is not aromatic in nature. In cyclooctatetraene, delocalisation of π− electrons takes place but HuckeFs rule is not followed.