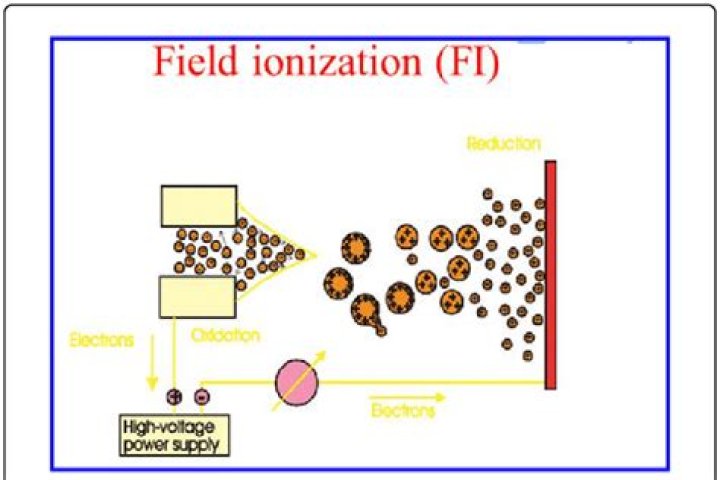
In field ionization, electrons are removed from a species by quantum mechanical tunneling in a high electric field, which results in the formation of molecular ions (M + ̇ in positive ion mode). This ionization method usually takes places in nonpolar or slightly polar organic compounds.
How does field ionization work?
In field ionization, electrons are removed from a species by quantum mechanical tunneling in a high electric field, which results in the formation of molecular ions (M + ̇ in positive ion mode). This ionization method usually takes places in nonpolar or slightly polar organic compounds.
What are the types of ionization?
- Atmospheric Pressure Chemical Ionization. …
- Atmospheric Pressure Photoionization (APPI) …
- Electrospray ionization (ESI) …
- Matrix-Assisted Laser Desorption Ionization (MALDI)
What are the two types of ionization?
There are many types of ionization methods are used in mass spectrometry methods. The classic methods that most chemists are familiar with are electron impact (EI) and Fast Atom Bombardment (FAB).Which of the technique involves electric field for the formation of molecular ion?
7.6. Field ionization mass spectrometry depends on the tunnelling of an electron from a molecule in the gas phase on passage through a steep electric field, to an electrode. … The molecule loses one electron in the electric field, to produce a molecular ion.
What is fab in mass spectrometry?
Fast atom bombardment (FAB) is an ionization technique used in mass spectrometry in which a beam of high energy atoms strikes a surface to create ions. … When a beam of high energy ions is used instead of atoms (as in secondary ion mass spectrometry), the method is known as liquid secondary ion mass spectrometry (LSIMS).
How is ionization achieved in field ionization?
A high electric field gradient (108 V/cm) at the tips of the whiskers produces ionization by quantum mechanical tunneling of electrons. FI is also used for performing isotope ratio measurements on samples that either give small molecular ions or large (M-H)+ ions in EI.
What creates ionization?
Ionization is the process by which ions are formed by gain or loss of an electron from an atom or molecule. If an atom or molecule gains an electron, it becomes negatively charged (an anion), and if it loses an electron, it becomes positively charged (a cation). Energy may be lost or gained in the formation of an ion.What is ionization with example?
Ionization is when an atom becomes ionized because it loses or gains an electron. … For example, chlorine can become ionized by gaining an electron to become negatively charged. Therefore, you can think of ionization as an atom going from a normal atom to an ion!
What causes ionization?Ionization, in general, occurs whenever sufficiently energetic charged particles or radiant energy travel through gases, liquids, or solids. … Pulses of radiant energy, such as X-ray and gamma-ray photons, can eject electrons from atoms by the photoelectric effect to cause ionization.
Article first time published onWhat ionised mean?
Ionization or ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule is called an ion.
What is the difference between oxidation and ionization?
Ionization energy is the amount of energy required to remove an electron. Oxidation potential is the opposite of the reduction potential, which is electrical potential (i.e., voltage) derived from comparing the spontaneity of the reduction compared to reducing a standard hydrogen electrode.
What are ionised gases?
Ionised gas is made of particles which have lost one or more electrons. This results in a positive charge of at least +1 on the ionised gas atom or molecule. Electrons may be lost due to a variety of reasons.
What is the difference between ESI and APCI?
CriteriaAPCIESIIonisationTypically, only generates singularly charged ions.Soft ionisation technique allows for the formation of multiply charged ions
How does electron ionization work?
Electron ionization occurs through interaction of the gas-phase sample molecules with high-energy electrons emitted from a resistively heated filament. After emission from the filament, the electrons are focused through a narrow slit and attracted to an anode that is positioned ~15–20 mm opposite the filament.
Why is ESI soft ionization?
ESI is a so-called ‘soft ionization’ technique, since there is very little fragmentation. This can be advantageous in the sense that the molecular ion (or more accurately a pseudo molecular ion) is almost always observed, however very little structural information can be gained from the simple mass spectrum obtained.
Who discovered the mass spectrometer?
The first mass spectrometer – originally called a parabola spectrograph – was constructed in 1912 by J.J. Thomson, best known for his discovery of the electron in 1897. He used the mass spectrometer to uncover the first evidence for the existence of nonradioactive isotopes.
How do the spectra for electron impact field ionization and chemical ionization differ from one another?
Electron impact ionization produces the most fragmentation and the most complex spectra. Field ionization produces the simplest spectra. Chemical and electron ionization produce greater sensitivities than does field ionization.
How do the spectra obtained with electron impact ionization differ from the spectra from chemical ionization?
Chemical ionization delivers less energy to the analytes and as a result causes less molecular fragmentation. … Compared to EI, this technique provides less energy resulting in “soft” ionization. Mass spectra acquired from a CI source generally contain the molecular ion.
Which of the following is not a MS detector?
Which of the following is not a type of ion detector used in mass spectrometers? Explanation: Flame emission detector is not a type of ion detector used in mass spectrometers.
How do alcohols fragment in mass spec?
In a mass spectrometer, alcohols fragment in two characteristic ways: alpha cleavage and dehydration. From the equation shown on page 657, you can see that the dehydration of an alcohol in a mass spectrometer is essentially the same as the dehydration of an alcohol in a normal chemical reaction.
How does electron bombardment?
Electron ionization (EI, formerly known as electron impact ionization and electron bombardment ionization) is an ionization method in which energetic electrons interact with solid or gas phase atoms or molecules to produce ions. EI was one of the first ionization techniques developed for mass spectrometry.
What is ionization PPT?
INTRODUCTION Ionization or ionisation, is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons to form ions, often in conjunction with other chemical changes.
What are the two ways to cause ionization?
Ionization can be induced by high energy radiation such as x rays and ultraviolet light (photoionization), bombardment by high energy electrons (electron impact ionization) or small molecular ions (chemical ionization) and by exposure to high electric fields (field ionization).
How do salts ionize?
The negative electrical charge is derived from electrons and the positive charge from protons in the nucleus. An example of an ion is sodium chloride (NaCl). When dissolved in water, sodium and chloride separate to form two kinds of ions: Na+ and Cl–. Sodium loses one electron and chloride gains one electron.
Can you ionize nitrogen?
Nitrogen cannot be ionized with standard ionization equipment designed for use in ambient air or CDA environments. The Model 4214 provides the user with the first small format ionizer that ionizes the actual nitrogen molecules instead of the trace impurity gases.
How do you make an ionizer?
- Step 1: Components and Design. …
- Step 2: Full-Wave Ionizer Materials and Component Layout. …
- Step 3: Half-Wave Ionizer Materials and Component Layout. …
- Step 4: Carbon Fibre Vs Pins for High Voltage Grid. …
- Step 5: Speed Wiring Tip. …
- Step 6: Full Wave Ionizer.
What happens if an atom is too ionized?
Ions are attracted to other atoms and molecules. If your atom is too ionized, it will likely zip away from you and attach itself to a nearby atom or molecule. An atom becomes radioactive when its nucleus contains too many or too few neutrons. Try to keep the same number of neutrons and protons as you build your atom.
What has the highest ionization energy?
Thus, helium has the largest first ionization energy, while francium has one of the lowest.
What is ionization in medicinal chemistry?
Ionization is the processby which an atom or a moleculeacquires a negativeor positive charge by gaining orlosing electronsto form ions, often in conjunctionwith otherchemicalchanges.
Is dissociation and ionization the same?
The key difference between ionization and dissociation is that dissociation is the process of separation of charged particles which already existed in the compound whereas ionization is the formation of new charged particles which were absent in the previous compound.