To sublimate is to change the form, but not the essence. Physically speaking, it means to transform solid to vapor; psychologically, it means changing the outlet, or means, of expression from something base and inappropriate to something more positive or acceptable.
What is a sublimation simple definition?
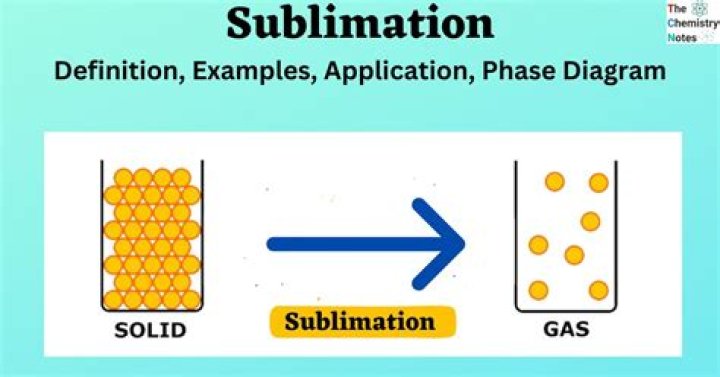
sublimation, in physics, conversion of a substance from the solid to the gaseous state without its becoming liquid. An example is the vaporization of frozen carbon dioxide (dry ice) at ordinary atmospheric pressure and temperature. The phenomenon is the result of vapour pressure and temperature relationships.
What is sublimation class 7th?
Answer: The term sublimation is the passage or the transformation or conversion that substances undergo when passing from one state to another, for example from a solid substance to gas. … Desublimation or deposition is the reverse of this process in which a gas is directly converted into solid-state.
What means Sublimable?
Filters. (chemistry) Capable of being sublimed or sublimated. adjective.What is sublimation in chemistry class 6?
Sublimation means substance directly converted in gaseous form. Those substances convert in gaseous form this is called as sublimate. Sublimation is also one kind of separation technique which we can use to separate solid-solid homogeneous mixture which is difficult to separate from one another.
What is sublimated jersey?
When a jersey is “sublimated”, the design, logos, numbers and player names do not fade, crack or wear, because they are directly infused into the fabric. Sublimation jerseys are different from screen-printed or embroidered jerseys. On such jerseys the logos, etc. … Over time the logos may wear or peel off.
What is sublimate in chemistry?
Sublimate is defined as to cause the change of a gas into a solid or a solid into a gas without becoming a liquid, or to have a refining effect on someone or something. An example of sublimate is dry ice turning directly into carbon dioxide from solid form. … (chemistry) A product obtained by sublimation.
What is sublimation in chemistry class 11?
Hint: Sublimation can be defined as the transition of a substance from solid to gaseous phase without forming the liquid phase.What are examples of sublimation?
- Dry ice is the solid form of Carbon Dioxide. …
- Ice crystals inside boxes of frozen foods: Frozen foods sublime and results in formation of ice crystals inside the box or packet.
Sublimation is the process in which a solid changes directly to a gas without going through the liquid state. Solid carbon dioxide is an example of a substance that undergoes sublimation.
Article first time published onWhat is sublimation according to class 9?
The term sublimation is the passage or the transformation or conversion that substances undergo when passing from one state to another, for example from a solid substance to gas. We can define sublimation as the transition of a substance from the solid phase to the gaseous phase without changing into the liquid phase.
What is Sublimation class 4th?
The term sublimation is the passage or the transformation or conversion that substances undergo when passing from one state to another, for example from a solid substance to gas. We can define sublimation as the transition of a substance from the solid phase to the gaseous phase without changing into the liquid phase.
What is Sublimation answer 6th class?
Answer: Sublimation is the change of the gaseous state directly into the solid state, without going through the liquid state, and vice versa.
What is Sublimation question and answer?
Answer: Sublimation is the transition of a substance directly from the solid to the gas phase without passing through the intermediate liquid phase. Answer: The process of conversion of solid to gas directly by absorbing heat without going through the liquid state is Sublimation .
What is sublimation in which lesson?
Lesson Summary Sublimation is a type of phase transition, or a change in a state of matter, just like melting, freezing, and evaporation. Through sublimation, a substance changes from a solid to a gas without ever passing through a liquid phase. Dry ice, solid CO2, provides a common example of sublimation.
What is sublimation and why does it occur?
The process in which a solid changes directly to a gas is called sublimation. It occurs when the particles of a solid absorb enough energy to completely overcome the force of attraction between them. … Solid carbon dioxide changes directly to the gaseous state.
What is a sublimated logo?
SUBLIMATED LOGOS: – Sublimation is the term used when digital print is transferred from printing paper to fabric under a very high heat process. This high heat will seal the colours into the fabric to ensure they do not run or fade.
What is sock sublimation?
The sublimation sock blanks are 100% white polyester with black detailing around the toe and the heel. Each pair is individually wrapped with a cardboard insert.
How do you use sublimation in a sentence?
- Rather than start an argument I chose to sublimate my anger by clenching my fists and walking away.
- The artist is known for his ability to sublimate his anger into his paintings.
- When Gail’s husband died, she tried to sublimate her grief by feeding the homeless.
What is sublimation and two example?
Examples for sublimation process Sublime snow and ice without melting during winter. … Moth balls sublime. Dry ice sublimes. Iodine, which sublimates from solid to poisonous purple gas at 100 degrees C.
What is mixture Class 9?
A mixture is a substance which consist of two or more elements or compounds not chemically combined together. For Example Air is a mixture of gases like oxygen,nitrogen,argon,carbon dioxide etc. … The various substance present in a mixture are called as its constituents or components.
What is fusion in chemistry class 9?
Fusion. The phenomenon of change of solid into liquid is termed as fusion. For instance, melting of ice.