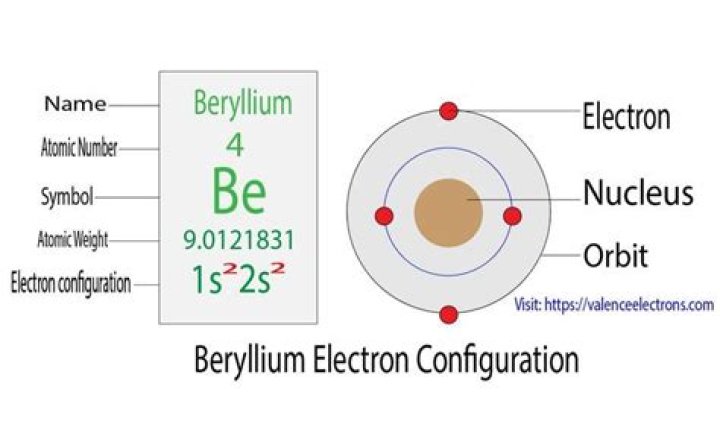
Beryllium is the fourth element with a total of 4 electrons. In writing the electron configuration for beryllium the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the remaining 2 electrons for Be go in the 2s orbital. Therefore the Be electron configuration will be 1s22s2.
What electron configuration does beryllium have?
Beryllium is the fourth element with a total of 4 electrons. In writing the electron configuration for beryllium the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the remaining 2 electrons for Be go in the 2s orbital. Therefore the Be electron configuration will be 1s22s2.
What bond order does O2 2 have?
The bond order for the O22- is 1 (one). In a neutral O2 molecule, there are a total of 12 valence shell electrons shared between the bonded atoms.
What is the electron configuration for beryllium atomic number 4?
ElementAtomic numberElectron configurationhydrogen11s1helium21s2lithium31s22s1beryllium41s22s2How do you write the electron configuration?
Writing Electron Configurations. When writing an electron configuration, first write the energy level (the period), then the subshell to be filled and the superscript, which is the number of electrons in that subshell. The total number of electrons is the atomic number, Z.
What is 1s 2s 2p 3s 3p?
1s 2s 2p 3s 3p represents the electron orbital energy levels.
What is the electronic configuration of N2?
The electronic configuration of N2 is (σ1s) 2 (σ∗1s) 2 (σ2s) 2 (σ∗2s) 2 (π2px) 2 (π2py) 2 (σ2pz) the total number of electrons present in the nitrogen molecule (N 2 ) is 14.
How do you write the electron configuration for lithium?
Lithium is the third element with a total of 3 electrons. In writing the electron configuration for lithium the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the remaining electron for Li goes in the 2s orbital. Therefore the Li electron configuration will be 1s22s1.What is the electron configuration for 39 19 K?
Group163.5°C, 146.3°F, 336.7 KBlocks0.89Atomic number1939.098State at 20°CSolid39KElectron configuration[Ar] 4s17440-09-7
What is the bond order of C2+?Bond order = 1/2 (number of electrons in bonding orbitals – number of electrons in antibonding orbitals) Therefore, Bond order of C2+ = 1/2 (5 – 2) = 3/2 = 1.5.
Article first time published onWhat is the bond order of N2+ N 2?
That is, the bond order for N2+ is 2.5.
What is the electron configuration 1s2 2s2 2p6?
ABCarbon1s2 2s2 2p4Nitrogen1s2 2s2 2p5Silicon1s2 2s2 2p6 3s2 3p2Phosphorous1s2 2s2 2p6 3s2 3p3
What is the electronic configuration of n2 negative?
The electronic configuration of nitrogen (Z=7) = 1s2 2s2 2px12py12pz1. The total number of electrons present in the nitrogen molecule (N2) is 14.
How are 15 electrons arranged in a phosphorus atom?
A neutral phosphorus atom has 15 electrons. Two electrons can go into the 1s subshell, 2 can go into the 2s subshell, and 6 can go into the 2p subshell. That leaves 5 electrons. Of those 5 electrons, 2 can go into the 3s subshell, and the remaining 3 electrons can go into the 3p subshell.
Where is 5f on the periodic table?
In order to make the overall shape of the table more compact and convenient to display, scientists have adopted the convention of removing the elements with atomic number 57 through 70 and 89 through 102 (the latter being the 5f portion of the table) from their natural position between the s and d blocks and placing …
What is the electron configuration order?
Aufbau Principle According to this principle, electrons are filled in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p…
What is 1s & 2s?
1s orbital is the closest orbital to the nucleus. 2s orbital is the second closest orbital to the nucleus. Energy Level. Energy of 1s orbital is lower than that of 2s orbital. 2s has comparatively higher energy.
What does the symbol 39 19 K mean?
It is the number under the element symbol. For potassium it is about 39. This means that the atomic weight is 39 for both protons and neutrons. Since we know that the number of protons is 19 we can calculate the number of neutrons (39 19) as 20.
How many 4s electrons are in K?
A. weight4.003NameHeliumSymbolHeGroup18Electron configuration1s2
How many protons neutrons and electrons are there in 40k?
19 protons, 20 neutrons and 18 electrons.
What is the effect of the process C2 → C2+ E on bond order of C2?
Bond order for C2 → C2+ e- decreases from 2 to 1.5 but the bond order for O2 → O2+ + e- increases from 2 to 2.5.
How do you find the bond order?
- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.
What is the bond order of N2 3?
Chemical Bonding and Molecular Structure. Give the reason for the following: Bond order in N2 is 3 whereas it is 2.5 in NO ? Explain the formation of H2 molecule on basis of valence bond theory.