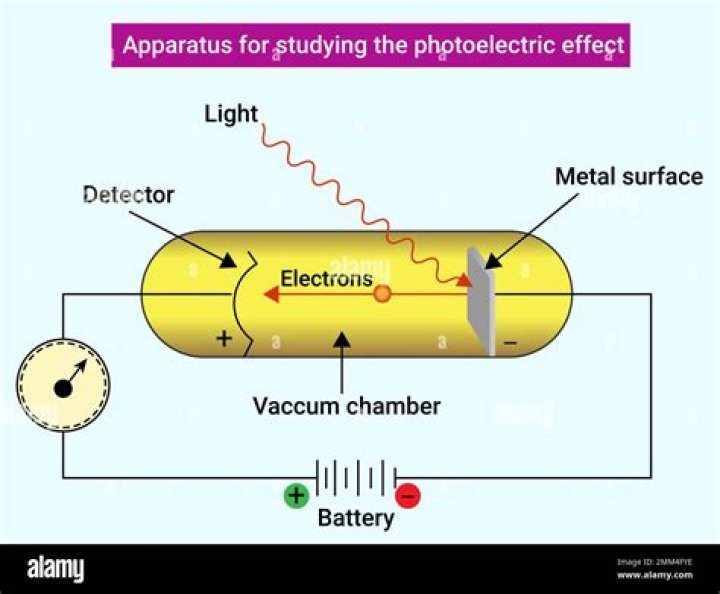
The type of light that is needed to cause the photoelectric effect is any light that has enough energy (photon energy) to facilitate the ejection of electrons from a metal. In most cases, it is the ultraviolet light.
What causes the photoelectric effect?
The photoelectric effect is a phenomenon that occurs when light shined onto a metal surface causes the ejection of electrons from that metal. It was observed that only certain frequencies of light are able to cause the ejection of electrons. … At or above the threshold frequency (green) electrons are ejected.
What is the requirement for the photoelectric effect?
The minimum energy required to eject an electron from the surface is called the photoelectric work function. The threshold for this element corresponds to a wavelength of 683 nm. Using this wavelength in the Planck relationship gives a photon energy of 1.82 eV.
Can visible light cause photoelectric effect?
The photoelectric effect is the process in which electromagnetic radiation such as visible light, x rays, or gamma rays strike matter and cause an electron to be ejected. The ejected electron is called a photoelectron.What is photoelectric effect and wave theory of light?
The photoelectric effect occurs when light shines on a metal. … Predictions of the wave theory of light: Light of any frequency will cause electrons to be emitted. The more intense the light the more kinetic energy the emitted electrons will have.
Can sodium be used for photoelectric effect?
A sodium photoelectric surface with work function 2.3eV is illuminated and emits electrons. The electrons travel toward a negatively charged cathode and complete a circuit. The potential difference supplied by the power supply is increased, and when it reaches 4.5V, no electrons reach the cathode.
How does intensity of light affect photoelectric effect?
In the photoelectric effect, light incident on the surface of a metal causes electrons to be ejected. … The intensity affects the number of electrons, and the frequency affects the kinetic energy of the emitted electrons.
Why alkali metals are sensitive to visible light?
alkali metals said to be most suitable for the photoelectric effect because the alkali metal have more and more tendency of loosing electrons and hence makes it easier for the photon to eject it out from the atom with the consumption of less energy.Why sodium is used in photoelectric effect?
The minimum photon energy at which electrons can stick out is the ‘work function. ‘ Usually, alkali metals such as sodium(Na) are often used in photoelectric effect experiments. This is because alkali metals’ work function is small, so it is easy to cause a photoelectric effect even with low-frequency visible light.
How does the frequency of light affect the release of photons?The higher the frequency, the more energy the photon has. Of course, a beam of light has many photons. This means that really intense red light (lots of photons, with slightly lower energy) can carry more power to a given area than less intense blue light (fewer photons with higher energy).
Article first time published onWhat are knocked off when light photons strike metal surfaces?
The photoelectric effect is the observation that under certain conditions, light striking a metal surface can cause electrons to be ejected. … Einstein explained the result be describing the light waves as packets of energy. He called the packets of energy photons.
What is the effect of intensity of light?
In photoelectric effect, on increasing the intensity of light, both the number of electrons emitted and kinetic energy of each of them get increased but photoelectric current remains unchanged.
What is the photon theory of light?
According to the photon theory of light, photons: behave like a particle and a wave, simultaneously. … carry energy and momentum, which are also related to the frequency (nu) and wavelength (lamdba) of the electromagnetic wave, as expressed by the equation E = h nu and p = h / lambda.
Which electrons are emitted in photoelectric effect?
The photoelectric effect is the emission of electrons when electromagnetic radiation, such as light, hits a material. Electrons emitted in this manner are called photoelectrons.
What are individual particles of light called?
The light particle conceived by Einstein is called a photon. The main point of his light quantum theory is the idea that light’s energy is related to its oscillation frequency (known as frequency in the case of radio waves). … Photons have energy equal to their oscillation frequency times Planck’s constant.
How does wavelength affect photoelectric effect?
In the photoelectric effect light which strikes a metal causes electrons to be emitted. … As the wavelength decreases for a specified metal, the speed (and thus the Kinetic Energy) of the emitted electrons increases.
What is threshold frequency for photoelectric effect?
The threshold frequency is defined as the minimum frequency of incident radiation below which the photoelectric emission is not possible completely.
What is light intensity?
Light intensity refers to the strength or amount of light produced by a specific lamp source. It is the measure of the wavelength-weighted power emitted by a light source.
How do you use e HV?
- a Photon’s Wavelength (λ) and energy E are linked through the following equation:
- E = hc/λ
- WHERE:
- h = Planck’s constant = 6.63 x 10 J s (Joules per second)
- v = frequency of the radiaiton.
- c= speed of light.
- E is inversely proportional to λ.
How do you find the kinetic energy of a photoelectric effect?
Definition: The Maximum Kinetic Energy of a Photoelectron given Wavelength. The maximum kinetic energy of a photoelectron is given by 𝐸 = ℎ 𝑐 𝜆 − 𝑊 , m a x where ℎ is the Planck constant, 𝑐 is the speed of light, 𝜆 is the wavelength of the incident photon, and 𝑊 is the work function of the metal surface.
What is H wavelength?
Wavelength is related to energy and frequency by E = hν = hc/λ, where E = energy, h = Planck’s constant, ν = frequency, c = the speed of light, and λ = wavelength.
Why is K lighter than sodium?
Potassium (K) is lighter than Sodium (Na) because of abnormal increase in the size of potassium. … But, in potassium the atomic radius increases highly whereas the mass is smaller in comparison to size and so the density of K decreases and it becomes lighter than Sodium Na.
Why is potassium and cesium used in photoelectric?
Potassium and caesium are used in photoelectric cells rather than lithium because potassium and caesium have much lower ionisation enthalpy than that of lithium. As a result, these two metals when subjected to light, emit electrons but lithium does not.
Why is cadmium used in photoelectric effect rather than lithium?
Answer Expert Verified Potessium and Caesium have much lower Ionisation enthalpy than that of Lithium . Therefore ,these metals on exposure to light emits electrons easily but lithium doesn’t . That’s why K and Cs rather than Li are used in photoelectric cells.
Which metals are suitable for photoelectric effect?
The work function of alkali metals is quite low (e.g. for potassium, it is 2.3 eV and for sodium is 2 eV). As a result, they show photoelectric effect even with visible light. For this reason, alkali metals are most suited for photoelectric emission.
Why does alkali metals show photoelectric effect?
Explanation: Alkali metals have low ionisation enthalpies and therefore, the electrons are easily ejected when exposed to light. Among alkali metals, cesium has lowest ionisation enthalpy and hence it can show photoelectric effect to the maximum extent.
Why are alkali metals most suitable for photoelectric effect?
Having only one electron in its valence shell. Alkali metals having very low ionization energy so it will be very easy to eject an electron. That’s why they are most suitable for photoelectric effect.
What determines the frequency of photons?
Each photon has a wavelength and a frequency. The wavelength is defined as the distance between two peaks of the electric field with the same vector. The frequency of a photon is defined as how many wavelengths a photon propagates each second.
How does the frequency of light affect the color of an object?
Color is determined first by frequency. … As the frequency is increased, the perceived color gradually changes from red to orange to yellow to green to blue to violet. The eye doesn’t perceive violet so well. It always seems to look dark compared to other sources at equal intensity.
What type of light on the electromagnetic spectrum has the highest energy per photon?
Gamma rays, a form of nuclear and cosmic EM radiation, can have the highest frequencies and, hence, the highest photon energies in the EM spectrum.
What causes blue light in photoelectric effect?
Each photon carries a specific energy related to its wavelength, such that photons of short wavelength (blue light) carry more energy than long wavelength (red light) photons.