As the temperature of a gas rises, the average velocity of the molecules will increase; a doubling of the temperature will increase this velocity by a factor of four. Collisions with the walls of the container will transfer more momentum, and thus more kinetic energy, to the walls.
What happens when an ideal gas molecule collides with its container walls?
Each time a gas particle hits the wall, it exerts a force on the wall. An increase in the number of gas particles in the container increases the frequency of collisions with the walls and therefore the pressure of the gas.
When molecules collide with the walls of their container What is exerted?
The pressure exerted by a gas in a container results from collisions between the gas molecules and the container walls. Gas molecules exert no attractive or repulsive forces on each other or the container walls; therefore, their collisions are elastic (do not involve a loss of energy).
What happens when ideal gas particles collide?
Gas particles are in a constant state of random motion and move in straight lines until they collide with another body. The collisions exhibited by gas particles are completely elastic; when two molecules collide, total kinetic energy is conserved.Do ideal gas molecules collide?
The term ideal gas refers to a hypothetical gas composed of molecules which follow a few rules: Ideal gas molecules do not attract or repel each other. The only interaction between ideal gas molecules would be an elastic collision upon impact with each other or an elastic collision with the walls of the container.
How do molecules collide?
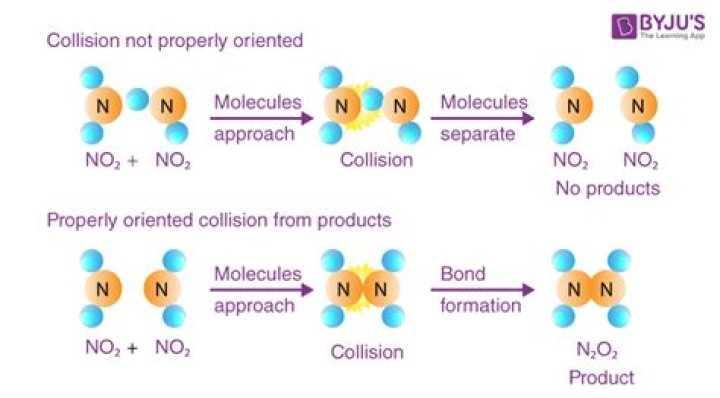
Molecules must collide with sufficient energy, known as the activation energy, so that chemical bonds can break. Molecules must collide with the proper orientation. A collision that meets these two criteria, and that results in a chemical reaction, is known as a successful collision or an effective collision.
Why does a collision with the wall of the container cause pressure?
Molecules of gas are always in random motion within the container. … The collisions result in the momentum exchange with the container wall & the rate of change of momentum results in the force exerted by gas molecule on the container wall. This force per unit area exerted by gas molecules is called pressure.
When molecules of a gas rebound from a wall of a container the wall experiences a pressure?
When a molecule collides with the wall of the container, a change of momentum occurs. The molecule exerts an equal but opposite force on the wall (Newton’s third law). The pressure exerted by the gas is due to the sum of all these collision forces.What do particles do in an ideal gas?
An ideal gas has identical particles of zero volume, with no intermolecular forces between them. The atoms or molecules in an ideal gas move at the same speed. Almost all gases obey the gas laws within a limited range of pressures and temperatures.
When gas molecules collide with each other there is a change in?Collisions are perfectly elastic; when two molecules collide, they change their directions and kinetic energies, but the total kinetic energy is conserved. Collisions are not “sticky”. The average kinetic energy of the gas molecules is directly proportional to the absolute temperature.
Article first time published onWhy does an ideal gas exert pressure on its container?
The pressure exerted by a gas is due to the random motion of particles in the gas. Gases have weak intermolecular forces and the particles are in continuous random motion and these particles collide with the walls of the container. These collisions with the walls of the container exert pressure on the gas.
What type of collisions do ideal gases have?
Elastic collision occurs between ideal gas molecules. Ideal gas molecules do not possess potential energy, i.e. they are not affected by intermolecular forces. The volume of ideal gas molecules is negligible compared to the volume of the container.
When air molecules collide with things around us what does it produce?
As the gas molecules collide with the walls of a container, the molecules impart momentum to the walls, producing a force that can be measured. The force divided by the area is defined to be the pressure. The temperature of a gas is a measure of the mean kinetic energy of the gas.
What is an ideal gas or perfect gas state equation of an ideal gas?
The ideal gas equation is formulated as: PV = nRT. In this equation, P refers to the pressure of the ideal gas, V is the volume of the ideal gas, n is the total amount of ideal gas that is measured in terms of moles, R is the universal gas constant, and T is the temperature.
What happens to the pressure of a gas inside a container if the amount of the gas decreases?
Volume. Pressure is also affected by the volume of the container. If the volume of a container is decreased, the gas molecules have less space in which to move around. As a result, they will strike the walls of the container more often, and the pressure increases.
How are collision and pressure related to each other?
Explanation: Molecules can collide with each other and with the walls of the container. Collisions with the walls account for the pressure of the gas. When collisions occur, the molecules lose no kinetic energy; that is, the collisions are said to be perfectly elastic. …
What does it mean when a collision is elastic?
An elastic collision is a collision in which there is no net loss in kinetic energy in the system as a result of the collision. Both momentum and kinetic energy are conserved quantities in elastic collisions. … They collide, bouncing off each other with no loss in speed.
How often do gas molecules collide?
Instead, it can take several minutes for us to detect an aroma because molecules are traveling in a medium with other gas molecules. Because gas molecules collide as often as 1010 times per second, changing direction and speed with each collision, they do not diffuse across a room in a straight line.
What happens when two atoms molecules collide?
In the first collision, the particles bounce off one another and no rearrangement of atoms has occurred. … The second collision occurs with greater kinetic energy, and so the bond between the two red atoms breaks.
What is it called when atoms collide?
an elementary collision event between two atomic particles, which may be atoms, molecules, electrons, or ions.
What happens when gas molecules collide at low temperatures?
At low temperatures, gas particles have less kinetic energy, and therefore move more slowly; at slower speeds, they are much more likely to interact (attracting or repelling one another) upon collision. The Ideal Gas Law does not account for these interactions.
Why do molecules move in a gas?
Gas In a gas, particles are in continual straight-line motion. The kinetic energy of the molecule is greater than the attractive force between them, thus they are much farther apart and move freely of each other.
Is a mixture of ideal gases also an ideal gas give an example?
Normally yes. Air, for example, behaves as an ideal gas in the range of temperatures and pressures at which oxygen and nitrogen behave as ideal gases.
What is the main process by which energy is released in the sun Igcse?
Nuclear fission and nuclear fusion Nuclear fusion, which occurs in the Sun, is the process in which two small nuclei combine to form a larger one, again releasing a huge amount of energy. When hydrogen atoms within stars combine to form helium the process is called fusion (see page 235).
Why does the pressure inside a cylinder fall when the temperature falls?
If the temperature decreases the pressure will also decrease. Temperature is a measure of kinetic energy. When the temperature inside the container decreases the kinetic energy will also decrease.
What causes the ripples to refract?
Shows refraction is caused when plane waves change speed as a result of a change in the depth of water in a ripple tank.
Do gas particles collide into each other?
Gas molecules influence each other only by collision; they exert no other forces on each other. They do not stick to each other. … All collisions between gas molecules are perfectly elastic; all kinetic energy is conserved.
What is kinetic theory of an ideal gas?
It says that the molecules of gas are in random motion and are continuously colliding with each other and with the walls of the container. All the collisions involved are elastic in nature due to which the total kinetic energy and the total momentum both are conserved. No energy is lost or gained from collisions.
How frequent do gas molecules hit the walls of the container as temperature changes?
If the temperature is increased, the average speed and kinetic energy of the gas molecules increase. If the volume is held constant, the increased speed of the gas molecules results in more frequent and more forceful collisions with the walls of the container, therefore increasing the pressure (Figure 1).
How does the kinetic theory explain a gas exerts a pressure on its container walls?
The concept of pressure is explained in kinetic theory as a consequence of kinetic energy of gases. … Due to the troublesome motion of the gas molecules, they will collide with each other, some of them shall collide with the container walls and then bounce back and this process continues.
What assumptions do chemists make about ideal gases What is ideal about ideal gases )?
The ideal gas law can be derived from the kinetic theory of gases and relies on the assumptions that (1) the gas consists of a large number of molecules, which are in random motion and obey Newton’s laws of motion; (2) the volume of the molecules is negligibly small compared to the volume occupied by the gas; and (3) …