H2O is more acidic than NH3 due to the more electronegative 0 present in H2O furthermore the lone pair of NH3 can easily be donated which makes it more basic than water.
Is NH3 a stronger base?
As it happens, ammonia (NH3) is not a strong acid; it is a weak base.
Is H2O a strong base?
Re: H2O – Weak Acid or Strong Base? Not exactly. Although we often think of water as being either a weak acid or base in reality it is a neutral compound as water has a pH of 7.
Which is a stronger base OH or H2O?
Why is hydroxide ion a stronger base than water? The oxygen in hydroxide ion is a better proton (H+) acceptor than the oxygen in water because the electron density at the oxygen in hyroxide ion is higher. The oxygen atom in hydroxide is sharing fewer of its electron pairs and is therefore more negative.Can H2O be a base?
Answers. Under the right conditions, H2O can donate a proton, making it a Brønsted-Lowry acid. Under the right conditions, H2O can accept a proton, making it a Brønsted-Lowry base.
Which is a stronger base nh3 or CH3NH2?
The CH3NH2 is more basic than ammonia. It is because the CH3NH2 has methyl group which is usually an election releasing group. It has +l effect of the methyl group. For this electrons are more concentrated over Hydrogen.
Which substance is the strongest base?
- Potassium hydroxide (KOH)
- Sodium hydroxide (NaOH)
- Barium hydroxide (Ba(OH)2)
- Caesium hydroxide (CsOH)
- Sodium hydroxide (NaOH)
- Strontium hydroxide (Sr(OH)2)
- Calcium hydroxide (Ca(OH)2)
- Lithium hydroxide (LiOH)
Is nh3 a weak base?
Ammonia is a typical weak base. Ammonia itself obviously doesn’t contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions.Which is stronger base H2O or Cl?
The general rules suggest that the stronger of a pair of acids must form the weaker of a pair of conjugate bases. The fact that HCl is a stronger acid than the H3O+ ion implies that the Cl- ion is a weaker base than water.
Is NO2 strong or weak base?But stronger acid implies weaker conjugate base and weaker acid implies stronger conjugate base. So,nitrite (NO2^-)is weaker base than sulphite(SO3^2-).
Article first time published onWhy is H2O a weak base?
Yes, pure water acts as a weak base. In fact, pure water acts as both a weak acid and a weak base. This is because a small amount of water dissociates into protons and hydroxide anions, binding with the remaining water molecules to form hydronium ions and hydroxide ions.
Is NH3 a Bronsted base?
the NH3 accepts a proton and is a Bronsted-Lowry base.
Is white vinegar a base or acid?
Vinegar is acidic. Vinegar’s pH level varies based upon the type of vinegar it is. White distilled vinegar, the kind best suited for household cleaning, typically has a pH of around 2.5.
Is bleach a base?
Chlorine bleach is a base and is especially good at removing stains and dyes from clothes as well as disinfecting.
Which is stronger base NH3 or PH3?
Due to smaller atomic size the density of lone pair electrons on N in NH3 is larger than that of P in PH3. So, NH3 is a stronger Lewis base than that of PH3.
Which is the weakest base?
Basic character of hydroxides of s-block elements increase with increasing atomic number. However alkaline earth metal hydroxides are less basic than alkali metal hydroxides. Therefore, Li(OH) is the weakest base.
Which of the following are stronger bases than NH3?
N2H4 is a stronger base than NH3 and N3H is a weaker acid than NH3COOH .
Which is more basic NH3 or nr3?
NH3 is more basic than NF3. … In NH3,N is more electronegative than H, hence bond pair shifts towards N in N-H, thereby negative charge density on N increases, hence tendency to donate electrons increases.
Which one of the following is the strongest base in aqueous solution?
Diethylamine is the strongest base in aqueous solution.
Is OH or NH3 a stronger base?
NH3 is more acidic, as it is amphoteric. This means it can either donate or accept a proton. OH on the other hand, only has one H+ atom, so it can only accept a proton, indicating basic properties.
Which ion is the strongest base quizlet?
Any acid stronger than hydronium ion will simply transfer its proton to a water molecule to form hydronium ions. – Hydroxide ion is the strongest base that can exist in water to any significant extent. Any base stronger than hydroxide will remove a proton from water to form hydroxide ions.
Is NH3 a conjugate acid or base?
NH3 and NH4+ are a conjugate acid-base pair. NH4+ is the conjugate acid of the base NH3.
Is NH3 a strong weak or Nonelectrolyte?
It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion.
Why is NH3 a weak alkali?
The aqueous solution of ammonia is weak alkali because ammonia produces OH- ions in it’s water solution. – Initially ammonia doesn’t have any kind of OH- or hydroxyl ions. – But when ammonia reacts with the water ,some amount of water is dissociated and forms OH- ions with the help of the mixed ammonia.
Is HClO3 strong acid?
The 7 common strong acids are: HCl, HBr, HI, HNO3, HClO3, HClO4 and H2SO4 (1st proton only). … Remember what we mean by a weak acid or weak base. They simply do not ionize completely whereas a strong acid or base does.
How do you tell if an acid or base is strong or weak?
Any acid that dissociates 100% into ions is called a strong acid. If it does not dissociate 100%, it is a weak acid.
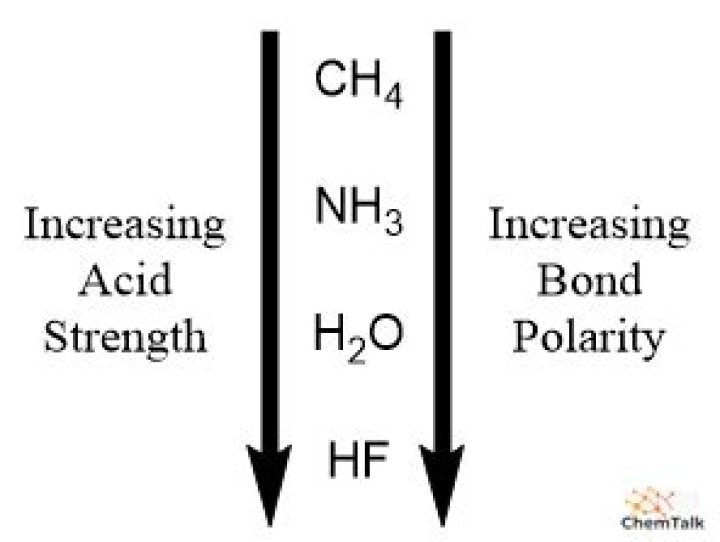
Why is H2O a stronger acid?
As electonegativity decreases down the group, oxygen is less electronegative than fluorine. So, compared to fluorine, it can easily loose hydrogen atom. As strength of acids is characterised by the ability to donate a hydrogen atom, hence H2O is stronger acid than HF.
Is NH3 H2O a base?
A substance that accepts and H+ from water is considered a base. Both NH3 and H2O are amphoteric (they have H atoms that can be donated as H+ ions and thus act as acids and lone-pair electrons that can accept an H+ and thus act as bases). Thus, either NH3 or H2O can act as an acid or a base.
Is h20 a conjugate base or acid?
BaseConjugate acidF − Fluoride ionHF Hydrogen fluoridePO 3− 4 Phosphate ionHPO 2− 4 Hydrogen phosphate ionOH− Hydroxide ionH2O Water (neutral, pH 7)HCO − 3 BicarbonateH 2CO 3 Carbonic acid
Why water H2O is considered a Brønsted-Lowry base in the reaction between water and HCl?
Because water accepts a proton from HClstart text, H, C, l, end text, water is acting as a Brønsted-Lowry base.
What is the pH of Coke?
How acidic is Coke? Its pH is reported to be 2.6 to 2.7, mainly due to H3PO4, phosphoric acid.