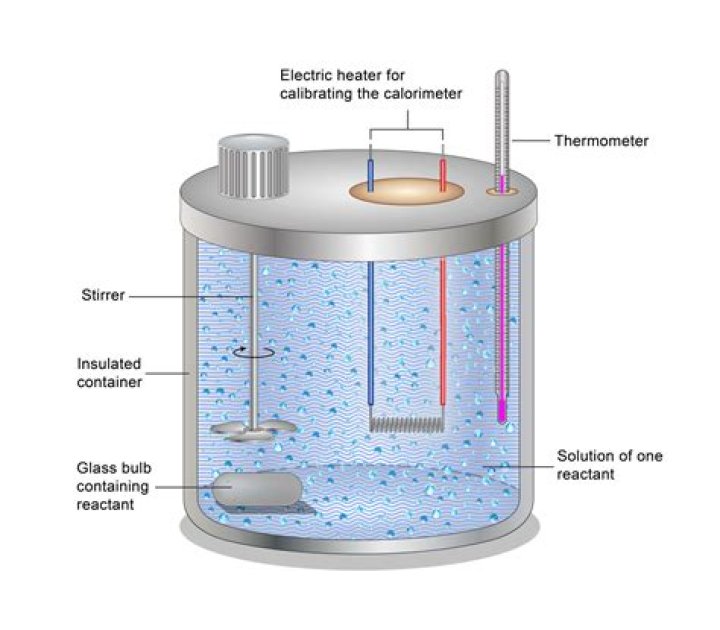
A calorimeter uses an insulated lid, or insulated surroundings. In the CAL2K the polystyrene surrounding is used as insulation to keep the heat of the liquid or solid sample during a reaction for an accurate measurement in the change of temperature to provide the best possible accurate results.
Why it is important to use an insulated closed vessel as a calorimeter?
Scientists use well-insulated calorimeters that all but prevent the transfer of heat between the calorimeter and its environment, which effectively limits the “surroundings” to the nonsystem components with the calorimeter (and the calorimeter itself).
What would happen if you forgot to place the lid on the calorimeter?
Suppose that during the experiment, a student forgot to put the lid on the calorimeter. What is the most likely error and reason? Temperature readings will be too high because heat will be absorbed by the air. Temperature readings will be too high because the lidless coffee cups will give off heat.
Why do you put a lid on the Styrofoam cup?
Coffee cups, especially those made of Styrofoam, are effective calorimeters because they hold in the heat. … The lid should be flat and make a good seal when placed inside the lip of the cup. Cardboard acts as a better insulator than plastic lids do.Why it is important to stir the water in the calorimeter?
Explanation: Well, so as to prevent hot-spots, and burning on the bottom of the pan. When you do a calorimetric experiment, you have a given mass of water, and you want the temperature rise of this mass to be uniform. So you stir it.
Is a calorimeter a closed system?
The calorimeter used is a closed system.
Why does a calorimeter have a double walled container?
E 14.1, the law of cooling apparatus has a double walled container, which can be closed by an insulating lid. Water filled between double walls ensures that the temperature of the environment surrounding the calorimeter remains constant.
Why was the calorimeter made from an insulating material like plastic foam?
They are insulated to prevent loss or gain of heat energy between the calorimeter and the surroundings.Why do calorimeters have two cups?
Relatively inexpensive calorimeters often consist of two thin-walled cups that are nested in a way that minimizes thermal contact during use, along with an insulated cover, handheld stirrer, and simple thermometer.
Why are two Styrofoam cups used in the calorimeter?The insulation of the styrofoam cups prevent any other heat (of course the insulation is not perfect), so the two heats must add to zero: Q1 + Q2 = 0 .
Article first time published onHow does a calorimeter prevent heat loss?
Answer: Heat loss due to conduction is prevented by placing the calorimeter box in a well-lagged vessel using wool or cork material. Heat loss due to convection is prevented by placing a lid on the box.
Why the copper cylinder of the calorimeter is placed in a wooden jacket?
Answer Expert Verified ▶To provide insulation its places in a wooden jacket and the space between wooden jacket and copper sheet is filled with wool or cotton to avoid heat by conduction. … ▶Copper have low specific heat capacity , so takes up small amount of heat from substance.
How does a calorimeter work?
A typical calorimeter works by simply capturing all the energy released (or absorbed) by a reaction in a water bath. … Thus by measuring the change in the temperature of the water we can quantify the heat (enthalpy) of the chemical reaction.
What if water bath is not stirred?
If you don’t stir the water, then the water bath is not in thermal equilibrium with itself… so different parts of it may be at different temperatures. You want the part of the water by the thermometer to be representative of all the water.
How do you stir a calorimeter?
Mechanically – Add a magnetic stirring bar to the calorimeter, clamp the calorimeter to a ring stand above a magnetic stirring plate, and turn the stirring plate to its lowest setting. Be careful, even low speed stirring can cause splashing, and may destroy the coffee cup.
Why do reactions need to be stirred?
Stirring keeps reactant particles in motion increasing the chances of collision and increasing the rate of reaction. Some reactions occur very slowly in the dark but much more quickly in light.
Why is a calorimeter insulated?
A calorimeter uses an insulated lid, or insulated surroundings. … The goal of insulating the bomb calorimeter system is to minimize the temperature loss from the system (calorimeter) to the surroundings (air) over time.
Does a calorimeter absorb heat?
Ideally, the components of the calorimeter would absorb no heat, but the components of the calorimeters always absorb some energy from the system. The heat capacity of the calorimeter is the quantity of heat absorbed by the calorimeter for each 1°C rise in temperature.
Why is it important to have enough water in the calorimeter to completely cover the metal sample?
Water has a high specific heat, which means it is difficult to increase the temperature of water. However, water also has the capacity to retain heat, which allows the other substance inside the calorimeter to absorb that heat.
Is a calorimeter open closed or isolated system?
Answer: An open system can exchange both matter and energy with the surroundings. ∆ H is measured when an open system is used as a calorimeter. A closed system has a fixed amount of matter, but it can exchange energy with the surroundings. … An isolated system has no contact with its surroundings.
What type of system is calorimeter?
A bomb calorimeter is a closed system because it allows heat to be exchanged.
What is the purpose of a calorimeter what kind of system open closed isolated Do we want a calorimeter to be why?
Because calorimetry is used to measure the heat of a reaction, it is a crucial part of thermodynamics. In order to measure the heat of a reaction, the reaction must be isolated so that no heat is lost to the environment. This is achieved by use of a calorimeter, which insulates the reaction to better contain heat.
Is a Styrofoam cup a perfect calorimeter?
A simple example of a constant-pressure calorimeter is a coffee-cup calorimeter, which is constructed from two nested Styrofoam cups and a lid with two holes, which allows for the insertion of a thermometer and a stirring rod. … As such, the outer cup is assumed to be a perfect insulator.
What is a Styrofoam calorimeter?
A coffee cup calorimeter is simply a styrofoam cup (or maybe one cup inside another) to provide insulation when materials are mixed inside of it. A styrofoam cover and a sensitive thermometer complete the apparatus.
Is Styrofoam an insulator?
Styrofoam has millions of small air bubbles trapped inside the foam. Since air is a bad conductor of heat Styrofoam efficiently prevents heat transfer. … Styrofoam reduces conduction and convection. In this way, it is one of the best existing insulators.
What makes an efficient calorimeter?
For a calorimeter to be effective it must have two properties. (1) Although any object will use or loose some heat in experiments, a calorimeter should not absorb / loose too much of heat, otherwise it would affect the readings and final results. … This is also the reason that calorimeters are properly insulated.
Is the calorimeter a good insulator?
A calorimeter is simply a container with insulating walls. … The Styrofoam coffee cup is a relatively good insulating material. A lid cardboard or other material also helps to prevent loss of heat, and a thermometer measures the change in temperature.
What material is the calorimeter made up of why?
Answer Expert Verified Calorimeter is made up of copper as copper has low specific heat capacity. 2. The molecules of liquids will have more kinetic energy than molecules of solid.
Why do we use a Styrofoam liner for the calorimeter?
A styrofoam cup is used because styrofoam is a relatively good insulating material. … To use the coffee cup calorimeter, one simply carries out the reaction to be studied inside the coffee cup, measures the temperature changes that take place, and then calculates the amount of heat lost or gained during the change.
What is bomb in bomb calorimeter?
The bomb calorimeter is used to determine the calorific values of solid and liquid fuels. It consists of a strong steel shell known as a bomb. It consists of a base which supports the platinum crucible and is screwed to the body of the bomb.
What type of heat transfer happens in a calorimeter?
… modes of heat transfer, i.e., convection, conduction, and radiation, are involved in the thermal-dynamic performance of a calorimeter, as shown in Fig. 2.