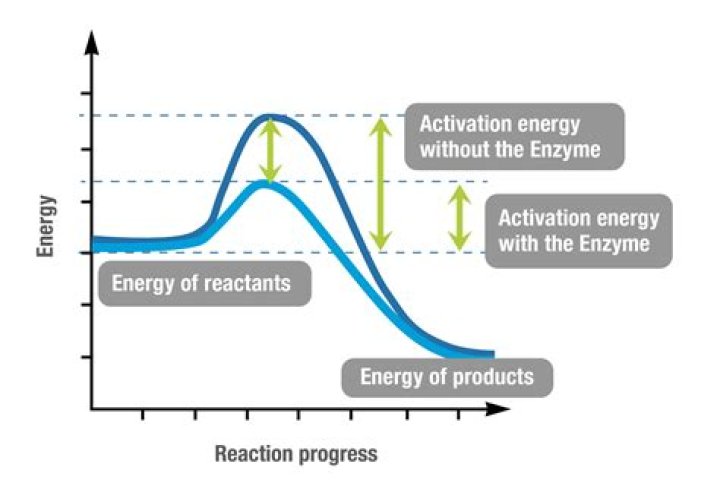
The activation energy is reduced so that the rate of reaction is faster. This is attained by adding enzymes to the reaction. Therefore, enzymes that act as catalysts speed up the reaction by reducing the activation energy.
Why does activation energy need to be lowered?
Enzymes lower the activation energy necessary to transform a reactant into a product. On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one that is (green). In the enzyme-catalyzed reaction, an enzyme will bind to a reactant and facilitate its transformation into a product.
Why is it important for enzymes to lower activation energy?
Compared to an uncatalyzed reaction (left), enzymes lower the activation energy by stabilizing the transition state into a more energetically favorable conformation (right). Enzymes facilitate chemical reactions by lowering the activation energy required for the reaction to occur.
Why is it important to lower down the activation energy of a chemical reaction?
Fortunately, it’s possible to lower the activation energy of a reaction, and to thereby increase reaction rate. The process of speeding up a reaction by reducing its activation energy is known as catalysis, and the factor that’s added to lower the activation energy is called a catalyst.What is lowering of activation energy?
Lowering the Activation Energy A catalyst is something that lowers the activation energy; in biology it is an enzyme. The catalyst speeds up the rate of reaction without being consumed; it does not change the initial reactants or the end products.
How does decreasing the concentration affect reaction rate?
When the concentration of all the reactants increases, more molecules or ions interact to form new compounds, and the rate of reaction increases. When the concentration of a reactant decreases, there are fewer of that molecule or ion present, and the rate of reaction decreases.
What is activation energy How is it lowered?
The source of the activation energy needed to push reactions forward is typically heat energy from the surroundings. For cellular reactions to occur fast enough over short time scales, their activation energies are lowered by molecules called catalysts. Enzymes are catalysts.
What slows down a chemical reaction?
The presence (and concentration/physical form) of a catalyst (or inhibitor). A catalyst speeds up a reaction, an inhibitor slows it down.Why would you want to slow down a chemical reaction?
A chemical reaction is taking place between two reactants dissolved in a liter of water. … We often want to decrease the rates of certain reactions rather than speeding them up. For example, to prolong the shelf lives of certain foods, the chemical reactions by which they spoil must be slowed down.
Why is it important that enzymes are not changed by the reactions?Enzymes lower the activation energy of the reaction but do not change the free energy of the reaction. … It is important to remember that enzymes do not change whether a reaction is exergonic (spontaneous) or endergonic. This is because they do not change the free energy of the reactants or products.
Article first time published onHow the active site of an enzyme can reduce the activation energy?
The enzyme-substrate complex can also lower activation energy by bending substrate molecules in a way that facilitates bond-breaking, helping to reach the transition state. Finally, some enzymes lower activation energies by taking part in the chemical reaction themselves.
Do enzymes change the free energy of a reaction?
An enzyme helps catalyze a reaction by decreasing the free energy of the transition state. As a result, more product will be made because more molecules will have the energy necessary for the reaction to occur and the reaction will occur at a faster rate.
Is the activation energy required for a chemical reaction were reduced what would happen to the rate of the reaction?
The amount of extra energy, the activation energy needed to get a reaction started, can be reduced. If less activation energy is needed, then more reactant molecules will have enough energy to make productive collisions, and the speed of the reaction will increase.
Can the activation energy be negative?
An elementary reaction can not have a negative activation energy: it must be zero or positive. However, a reaction mechanism that is composed of several steps may have a negative activation energy. … Negative activation energy is possible even for elementary reactions.
Does activation energy change with temperature?
The minimum energy needed for a reaction to proceed, known as the activation energy, stays the same with increasing temperature.
How does activation energy affect the position of equilibrium?
For an equilibrium chemical reaction to reach equilibrium, it requires some time. Equilibrium reactions with smaller activation energies will reach the state of chemical equilibrium in less time than reactions requiring higher activation energies.
Which of the following will lower the activation energy for a reaction?
Catalyst is a chemical species that fastens up the rate of the reaction by lowering the activation energy of the reaction and can be recycled at the end of the reaction. Catalysts were not consumed during the reaction. Therefore, option B is the correct answer.
Does breaking a reactant down into smaller pieces increase or decrease the rate of reaction?
Breaking the reactant into smaller pieces increases the surface and more particles are exposed to the reaction mixture. This results in an increased frequency of collisions and therefore a faster rate of reaction.
Why does concentration affect reaction rate?
Increasing the concentration of one or more reactants will often increase the rate of reaction. This occurs because a higher concentration of a reactant will lead to more collisions of that reactant in a specific time period.
Why are reactions faster at the beginning and then slow down?
This is largely to do with how much of the reactants are present. … As a result there will be more successful collisions between the reactant particles. The more successful collisions, the faster the reaction will be.
What are three ways to slow down a chemical reaction?
- temperature.
- stirring.
- changing the size of the reactants.
- changing the concentration of the reactants.
- add/remove substance.
- catalyst.
- inhibitors.
How can the rate of a reaction be reduced?
Perhaps the most obvious answer is to decrease the temperature of the reaction: this slows down the particles and, thus, reduces the likelihood of successful collisions between the reactant particles.
What should be done to slow down the rate of chemical change in an iron made material?
Galvanize: Galvanizing coats iron or steel in zinc to protect from rust. Zinc corrodes at a much slower rate than iron or steel, so it’s highly effective for slowing rust.
How does an enzyme change the activation energy of the reaction does it increase decrease or remain the same?
enzymes lower the activation energy for chemical reactions. enzymes change the Keq for chemical reactions. many enzymes change shape slightly when substrate binds. reactions occur at the “active site” of enzymes, where a precise 3D orientation of amino acids is an important feature of catalysis.
Why do you think it is an advantage for the enzyme to remain unchanged while catalyzing a chemical reaction?
Why do you think it is an advantage for the enzyme to remain unchanged while catalyzing a chemical reaction? … The enzyme is not a direct reactant in the cellular chemical reaction and doesn’t change in order to avoid interference with cellular reactions.
How energy changes affect how easily a chemical reaction will occur?
How do energy changes affect whether a chemical reaction will occur? … Chemical reactions that absorb energy will not occur without a source of energy. Energy Changes. Energy is released or absorbed whenever chemical bonds are formed or broken during chemical reactions.
Why do enzymes lower activation energy quizlet?
–Enzymes tightly control the chemistry of living cells because they have the ability to LOWER the ACTIVATION barriers for specific reactions. … -An enzyme binds tightly to one or more molecules and holds them in a precise configuration that LOWERS the ACTIVATION ENERGY.
Which of the following is one way that enzymes decrease activation energy?
Which of the following is one way that enzymes decrease activation energy? –Enzymes denature, allowing more physical space for reactants to bind.
Why is the barrier of the activation energy beneficial for cells?
Why is the barrier of the activation energy beneficial for cells? Energy is stored in the chemical bonds of a cell’s organic molecules. The activation energy barrier prevents these molecules from spontaneously breaking down and releasing that energy.
Does activation energy affect Gibbs free energy?
Activation energy is the smallest amount of energy that must be inputed into the reaction to get it going. Whereas Gibbs free energy determines where the equilibrium will settle out and how much products are produced at the end of the reaction, the activation energy determines the rate at which the reaction ouccrs.
How are free energy and activation energy affected by an enzyme?
Enzymes decrease the Gibbs free energy of activation, but they have no effect on the free energy of reaction. … The free energy of the product is the same as it would be without the enzyme. Thus, the enzyme does not affect the free energy of the reaction.