Because today’s aspirin is mass produced in tablet form, Spectrophotometers are a reliable and economical way to keep the purity of Aspirin consistent throughout the manufacturing process. The purity and amount of acetylsalicylic acid in aspirin can be measured using a Visual Spectrophotometer.
What makes aspirin pure?
The purity of acetylsalicylic acid was determined by melting point. In this experiment the melting range for acetylsalicylic acid is 144°C-172°C which is higher than commercial aspirin which is132. 8°C-138.9°C, meaning that acetylsalicylic acid obtained in this experiment contained impurities.
Why is it important to determine the purity of aspirin?
It is important to ensure that the purified product is actually Aspirin. This is because if it is not Aspirin, then testing the purity of it will become useless. It is possible for the Aspirin to be converted back into salicylic acid due to hydrolysis.
How can the aspirin be purified and how could you check its purity?
The solid aspirin will be collected using vacuum filtration. Any other reaction ingredients that are soluble (this includes acetic acid, phosphoric acid, and water) will pass through the filter paper. The collected aspirin will be tested for its purity using FeCl3 (aq).Is aspirin pure or impure?
The compound acetylsalicylic acid (the active ingredient in aspirin) is a substance because it is a chemical compound that cannot be further reduced to other materials by physical means. However, the aspirin that contains the acetylsalicylic acid is a mixture.
What is the percentage purity of aspirin?
The aspirin was 99.6 % pure.
How can I get pure aspirin?
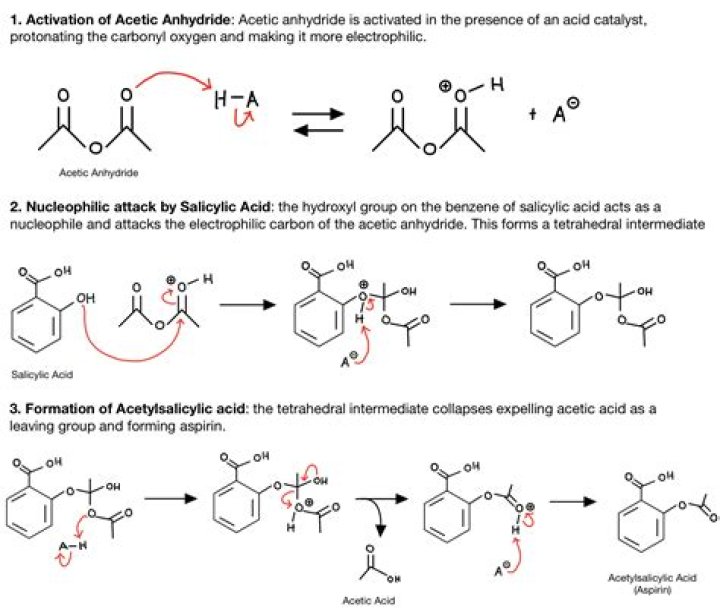
To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride. A small amount of a strong acid is used as a catalyst which speeds up the reaction. In this experiment, phosphoric acid will be used as the catalyst. The excess acetic acid will be quenched with the addition of water.
How do you test for aspirin?
Acetylsalicylic acid (ASA) is still one of the most commonly used therapeutic agents. Salicylic acid, the major metabolite of ASA, can be detected easily in urine using simple chemical spot tests such as ferric chloride or Trinder’s reagent.How does titration determine purity of aspirin?
Add several drops of phenolphthalein indicator. Slowly titrate the aspirin with the standardized NaOH solution. Record the initial and final buret readings to the correct number of significant figures. Calculate the percent purity of your sample.
How is aspirin measured?The standard adult aspirin dose was 5 gr, or 325 mg in metric, the dose still used today for analgesia. Low-dose aspirin was one quarter of the standard dose, 1.25 grains, which converted to 81 mg.
Article first time published onWhat impurities are in aspirin?
- 2-(Acetyloxy)benzoic Acid 4-(Chloromethyl)phenyl Ester. CAS No. …
- 3-Acetoxybenzoic Acid. CAS No. …
- 4-(2-Acetoxybenzoyloxy)benzaldehyde. CAS No. …
- 4-Hydroxyisophthalic Acid. CAS No. …
- Acetylsalicylic Anhydride. CAS No. …
- Acetylsalicylsalicylic Acid. CAS No. …
- Aspirin; Acetylsalicylic Acid. CAS No. …
- Benorilate. CAS No.
What impurities can be present in the synthesized aspirin?
The aspirin produced in this activity is not pure. It is often contaminated with salicylic acid, acetic acid, and/or sulfuric acid. Students should not taste their aspirin.
What are the synthetic and organic aspirins?
Aspirin (acetylsalicylic acid) is a synthetic organic derived from salicylic acid. Salicylic acid is a natural product found in the bark of the willow tree and was used by the ancient Greeks and Native Americans, among others, to counter fever and pain. However, salicylic acid is bitter and irritates the stomach.
How is aspirin Synthesised?
Aspirin is prepared by chemical synthesis from salicylic acid, through acetylation with acetic anhydride. The molecular weight of aspirin is 180.16g/mol. It is odourless, colourless to white crystals or crystalline powder.
What element is in aspirin?
The chemical name of aspirin is acetylsalicylic acid. By looking at its chemical structure, you’ll see that it’s composed of three different types of atoms: carbon, hydrogen, and oxygen. These atoms are either single or double bonded together to form the overall structure of aspirin.
What does acetylsalicylic acid look like?
Aspirin, an acetyl derivative of salicylic acid, is a white, crystalline, weakly acidic substance, with a melting point of 136 °C (277 °F), and a boiling point of 140 °C (284 °F).
What makes aspirin soluble?
Aspirin contains polar functional groups which can form hydrogen bonds with polar water molecules. … Ionic salts of aspirin, such as sodium acetylsalicylate, are more soluble in water since they form stronger ion-dipole interactions with water.
Why do you wash aspirin with cold water?
You cool the mixture because aspirin is still slightly soluble in water. You reduce the solubility of the product by lowering the temperature of the solvent. So cooling it is to maximize your yield.
How does titration determine purity?
Conversely, the percent purity of an impure sample of a chemical of unknown percent purity can be determined by reaction with a pure compound as in an acid-base titration. Percent purity can also be determined, in theory, by measuring the amount of product obtained from a reaction.
How much pure aspirin is in an aspirin tablet?
Each tablet contains 75 mg acetylsalicylic acid.
How do you find percent purity questions?
Percentage purity of a substance can be calculated by dividing the mass of the pure chemical by the total mass of the sample, and then multiplying this number by 100.
How do you find the percent purity of sodium hydroxide?
Percentage purity is the percentage of material actually useful or desired that is present in the sample. It can be calculated by the formula : $\frac{\text{weight of pure compound in sample}}{\text{total weight of impure sample}}\times 100$.
How do you determine concentration?
Divide the mass of the solute by the total volume of the solution. Write out the equation C = m/V, where m is the mass of the solute and V is the total volume of the solution. Plug in the values you found for the mass and volume, and divide them to find the concentration of your solution.
How do you measure aspirin in a tablet?
6. Calculate the mass of aspirin in each tablet according to the equation below. (MW of aspirin = 180 g/mole) Moles of aspirin × Molecular Weight of aspirin (in g/mole) = Mass of aspirin (in grams) 7. Record the mass of aspirin in each tablet (as claimed on the bottle) in Table 3.
Can aspirin cause a false positive?
Multivariate analysis found no association between regular aspirin or NSAID use and a false-positive test result (odds ratio = 0.85; 95% confidence interval: 0.39 to 1.84). Conclusion: Aspirin and NSAID use were not risk factors for a false-positive fecal occult blood test result in this study.
Which Colour is produced in identification test of aspirin?
Colour Test: Heat the sample with water for several minutes, cool, and add 1 or 2 drops of ferric chloride TS: a violet-red color is produced.
How long is aspirin detectable in blood?
The plasma half-life of aspirin is only 20 min in circulating blood. It is rapidly deacetylated and converted to salicylate in vivo. Salicylate does not affect COX-1 or COX-2 activity [3]. Because platelets cannot generate new COX, the effects of aspirin last for the duration of the life of the platelet [10 days].
What color is aspirin?
Colorless to white, crystalline powder.
How can you chemically detect the breakdown products of aspirin?
Analysis of Decomposition Products in Aspirin by Application of Two-wavelength Detection. The Chromaster 5410 (UV Detector) permits two-wavelength simultaneous measurements by realtime wavelength-switching.
Why does old aspirin smell like vinegar?
When acetylsalicylic acid ages, it may decompose and return to salicylic acid and acetic acid. If you have a very old bottle of aspirin around the house, open it and take a sniff. It may smell like vinegar, because vinegar is dilute acetic acid.
Why is aspirin better than salicylic acid?
Aspirin may exert its antiinflammatory activity mainly as salicylic acid which is not an inhibitor of prostaglandins biosynthesis in vitro. Aspirin showed about 5 times more potent analgesic action than sodium salicylate in the lameness test using adjuvant arthritic rats.