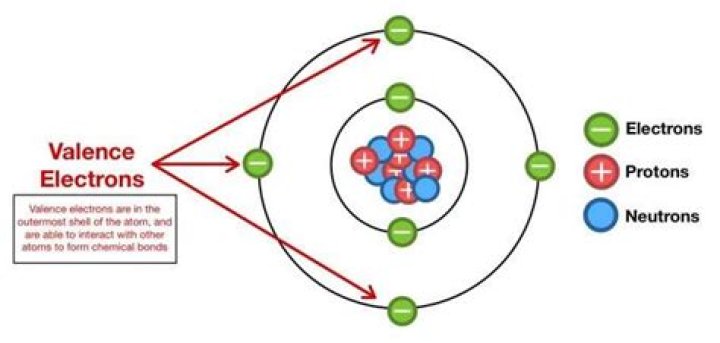
Ammonia also has four electron pairs and the coordination geometry of nitrogen is based upon a tetrahedral arrangement of electron pairs. There are just three bonded groups, therefore there is one lone pair.
How many electron groups does NH4+ have?
Ammonia also has four electron pairs and the coordination geometry of nitrogen is based upon a tetrahedral arrangement of electron pairs. There are just three bonded groups, therefore there is one lone pair.
Does NH4 have Valency?
The symbol for ammonium ion is NH4+ . It’s valency is +1 . It doesn’t exists in atomic form but exists in ionic form.
How many valence electrons does nh2 have?
NH2- Lewis Structure The Lewis structure for NH2- you have a total of 8 valence electrons.How many valence electrons does BF4 have?
In the Lewis structure of BF4- there are a total of 32 valence electrons. In the Lewis structure for BF4- Boron is the least electronegative atom and goes at the center of the structure.
What is the electron dot structure of NH4?
Answer: NH4+ is known as an ammonium ion, but NH4 would not make sense as it is not balanced. The lewis structure would be the Nitrogen atom in the center, with 4 hydrogens surrounding it. There would be no lone pairs, the molecule would also be nonpolar.
How many bonds does NH4+ have?
Three covalent bonds and one ionic bond.
What is the bond angle for NH4+?
Name of MoleculeAmmonium ionElectron geometry of NH4+TetrahedralHybridization of NH4+sp3Bond angle (H-N-H)109.5º degreeTotal Valence electron for NH4+ ion8What is the charge of NH4?
Globally, therefore, the NH4 molecule is positively charged, i.e., it is a cation. This particular cation is called the ammonium ion. Notice that +1 and –1 formal charges in HN3 balance each other out. Overall, the molecule is electrostatically neutral.
What is the hybridization of NH4+?Q. In the ammonium ion NH4+, what percentage of p character do the hybrid orbitals of nitrogen possess? The nitrogen atom forms 4 bonds and has 0 lone pairs, so it needs 4 hybrid orbitals. Its hybridization state is therefore sp3.
Article first time published onHow do you calculate ammonium valency?
The valency is almost always +1 as it has one electron free on the central N atom. The chemical formula of ammonium ion is [NH4]+. The valency is almost always +1 as it has one electron free on the central N atom.
How do I calculate bond order?
- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.
What is the shape of bf4?
Since all the valence electrons on Boron participate in bonding, there will be no lone-pair electrons on Boron. Hence the shape of \[B{F_4}^ – \] will be tetrahedral.
Does NH4+ have coordinate bond?
Three covalent bonds and one coordinate bond.
Does NH4 contain coordinate bond?
Complete answer: $N{H_4}^ + $contains three covalent and one coordinate bond. A coordinate bond which is also known as a dative covalent bond and dipolar bond is a type of two- centered and two- electron covalent bond where both electrons come from the same or single atom.
Is there coordinate bond in NH4?
The electrons of nitrogen have been denoted by dot and those of hydrogen atoms by the cross. So by definition, there are three covalent N-H bonds and one N → H coordinate bond in NH4+ ion.
How many electrons does nh3 have?
The nitrogen atom has 5 electrons and each of the three hydrogen atoms has 1 electron. The total number of electrons for ammonia will therefore be 8 electrons.
How many lone pairs does NH4 have?
After determining the center atom and sketch of NH4+ ion, we can start to mark lone pairs on atoms. Remember that, there are total of four electron pairs.
How many neutrons does NH4+ have?
In N atom there are 7 electrons, 7 protons and 7 neutrons.
Which ions are in NH4 2o?
The compound (NH4)2S ( N H 4 ) 2 S is formed from the cation ammonium (NH+4 N H 4 + ) and the sulfide anion (S2− ). Compounds made from cations and anions are ionic compounds.
Is NH4 a 2S?
Ammonium sulfide ((NH4)2S) exhibits high reactivity as a sulfide reagent in anion exchange reactions that transform CoO to cobalt sulfide nanoparticles (NPs).
What shape is the ammonium ion NH4+?
The ammonium ion has exactly the same shape as methane, because it has exactly the same electronic arrangement. NH4+ is tetrahedral.
What is the molecular geometry of NH4+ quizlet?
Ammonium, NH4+, has a central nitrogen atom surrounded by four hydrogen atoms. It has a tetrahedral electron geometry and a tetrahedral molecular shape.
What is the bond angle of ph3?
Name of the MoleculePhosphineMolecular FormulaPH3Hybridization TypeNilBond Angle93oGeometryTrigonal Pyramidal
What is the valency of NH4 so4?
NH4 is ammonium ion. So, valaency of ammonium ion is +1.
Why does ammonium only have 10 electrons?
In an ammonium ion (NH+4) ion, there is one nitrogen atom and four hydrogen atoms. Nitrogen has an atomic number of 7 , and hydrogen has an atomic number of 1 , and so the total atomic number of the hydrogens will be 1⋅4=4 . So, there will be a total of 7+4=11 protons. … So, there will be 10 electrons in an ammonium ion.
What is Silver's valency?
The most common valency of silver metal is 1. The symbol of silver is Ag.
Is NH4 ammonium?
NH4+ is the ammonium ion. It has a positive charge and a molecular weight of 18g/mol. NH3-N represents the Nitrogen content of the ammonia, NH4-N is the nitrogen content of the ammonium ion.
What is Chlorines Valence?
Chlorine has seven valence electrons and can form only one bond with an atom that donates a valence electron to complete chlorine’s outer shell. However, chlorine can also have oxidation states from +1 to +7 and can form more than one bond by donating valence electrons.
How was the no bond order of 4 3 calculated for no3?
For example, for NO3-, you have three bonds: One double bond (2 electron pairs) and two single bonds (1 + 1= 2 electron pairs). The bond order is therefore 4/3 = 1.33.
What does bond order 2.5 mean?
According to MOT, the bond order of is 2.5 & its electronic configuration is: . … The fact that NO has a bond order of 2.5 means that the bonding in NO is weaker than the N-N bond in N2. In MO theory, bond order is half the difference between the numbers of electrons in the bonding and antibonding molecular orbitals.