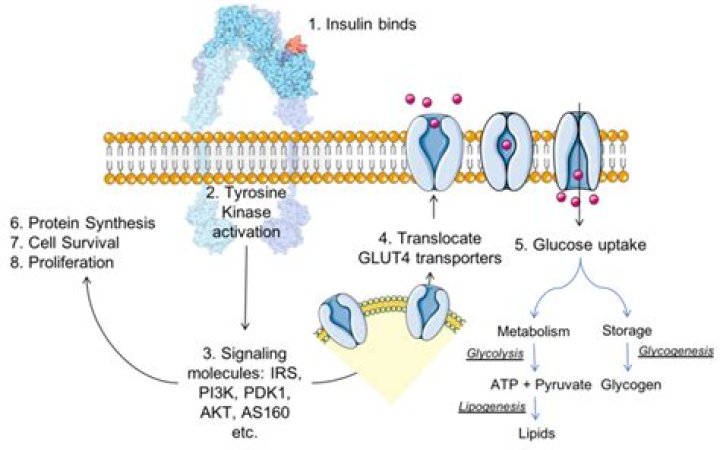
The insulin receptor is a member of the ligand-activated receptor and tyrosine kinase family of transmembrane signaling proteins that collectively are fundamentally important regulators of cell differentiation, growth, and metabolism.
Are insulin receptors tyrosine kinase receptors?
Signal transduction pathway The Insulin Receptor is a type of tyrosine kinase receptor, in which the binding of an agonistic ligand triggers autophosphorylation of the tyrosine residues, with each subunit phosphorylating its partner.
What type of receptor is tyrosine kinase?
Receptor tyrosine kinases (RTKs) are enzyme-linked receptors localized at the plasma membrane containing an extracellular ligand-binding domain, a transmembrane domain, and an intracellular protein–tyrosine kinase domain.
Does insulin activation tyrosine kinase?
The ligand (insulin) binds to IR, a receptor tyrosine kinase. Conformational changes resulting from insulin:IR binding activates the tyrosine kinase catalytic domain, which phosphorylates specific tyrosine residue found within the juxtamembrane and Tyr-K domains of the IR.What are insulin receptors made of?
Insulin receptor (IR) is a heterotetramer composed of two extracellular α-subunits and two transmembrane β-subunits, bound together by disulfide bonds.
Where does insulin bind insulin receptors?
Surprisingly, insulin binds to the outer edge of the receptor, and typically only binds to one side of the symmetrical receptor.
How does insulin work tyrosine kinase?
Insulin activates the insulin receptor tyrosine kinase (IR), which phosphorylates and recruits different substrate adaptors such as the IRS family of proteins. … Insulin stimulates glucose uptake in muscle and adipocytes via translocation of GLUT4 vesicles to the plasma membrane.
What is insulin receptor kinase activity?
Pathway Description: Insulin activates the insulin receptor tyrosine kinase (IR), which phosphorylates and recruits different substrate adaptors such as the IRS family of proteins. … Insulin stimulates glucose uptake in muscle and adipocytes via translocation of GLUT4 vesicles to the plasma membrane.What type of receptor is an insulin receptor?
The insulin receptor is a member of the ligand-activated receptor and tyrosine kinase family of transmembrane signaling proteins that collectively are fundamentally important regulators of cell differentiation, growth, and metabolism.
Is insulin a glycoprotein?Insulin initiates its cellular responses by binding to its cellular receptor, a transmembrane, multisubunit glycoprotein that contains insulin-stimulated tyrosine kinase activity [1].
Article first time published onWhere are receptor tyrosine kinases located?
A receptor tyrosine kinase (RTK) is a tyrosine kinase located at the cellular membrane and is activated by binding of a ligand via its extracellular domain.
What is a tyrosine kinase domain?
Tyrosine kinases are enzymes that selectively phosphorylates tyrosine residue in different substrates. Receptor tyrosine kinases are activated by ligand binding to their extracellular domain. Ligands are extracellular signal molecules (e.g. EGF, PDGF etc) that induce receptor dimerization (except Insulin receptor).
Is EGFR a tyrosine kinase?
The epidermal growth factor receptor (EGFR) is a receptor tyrosine kinase receptor that is frequently expressed in epithelial tumors. The EGFR was the first receptor to be proposed as a target for cancer therapy, and after 2 decades of intensive research, there are several anti-EGFR agents available in the clinic.
Is an insulin receptor an enzyme?
Following the demonstration in 1982 by Ora Rosen’s group that a tyrosine kinase was closely associated with the insulin receptor (14), several groups showed that the insulin receptor itself is a tyrosine kinase, an enzyme that catalyses the transfer of the g phosphate of ATP to tyrosine residues on protein substrates, …
Is insulin AG protein coupled receptor?
At the interface between these circulating factors and insulin/glucagon secretion are GPCRs, which in islets mediate the effects of many of the circulating factors, such as glucagon-like peptide-1, free fatty acids, and catecholamines.
Do all tissues have insulin receptors?
However, the existence of insulin receptors has been demonstrated in almost all tissues studied. Furthermore, certain tissues such as skeletal muscle and adipose tissue revealed the existence of insulin receptors despite the difficulty of morphological demonstration of insulin receptors in these tissues.
Is tyrosine kinase A protein?
Tyrosine kinases belong to a larger class of enzymes known as protein kinases which also attach phosphates to other amino acids such as serine and threonine.
How are insulin receptors activated after insulin binding?
Abstract. Insulin binding to insulin receptor (IR) at the cell surface results in the activation of IR kinase and initiates the translocation of insulin–IR complexes to clathrin-coated pits and to early endosomes containing internalized but still active receptors.
How does insulin activate a protein kinase?
Insulin activates a tyrosine-specific cAMP-independent protein kinase when added directly to detergent extracts of differentiated 3T3-L1 adipocytes and humal placental membranes. The kinase is also activated by antibody to the insulin receptor and, to a lesser extent, by proinsulin.
How does insulin cascade works?
When insulin binds to the insulin receptor, it leads to a cascade of cellular processes that promote the usage or, in some cases, the storage of glucose in the cell. The effects of insulin vary depending on the tissue involved, e.g., insulin is most important in the uptake of glucose by muscle and adipose tissue.
How does insulin work on a molecular level?
A Molecular Messenger Insulin is made in the pancreas and added to the blood after meals when sugar levels are high. This signal then spreads throughout the body, binding to insulin receptors on the surface of liver, muscle and fat cells.
Are insulin receptors synthesized on free or bound ribosomes?
Which proteins are synthesized by bound ribosomes? Proteins that function within the endomembrane system (such as lysosomal enzymes) or those that are destined for secretion from the cell (such as insulin) are synthesized by bound ribosomes.
Why are insulin receptors on alpha cells?
now provide evidence that pancreatic α cell insulin receptors modulate glucagon secretion in vivo. Their results highlight an important role of intraislet insulin signaling for control of glucagon secretion and glucose homeostasis.
How many receptor tyrosine kinases are there?
Of the 90 tyrosine kinase genes, 58 are of the receptor type as defined by encoding a protein with a predicted transmembrane domain. These 58 receptor tyrosine kinases can be grouped into 20 subfamilies based on kinase domain sequence.
How are receptor tyrosine kinases activated?
Generally, RTKs are activated through ligand-induced oligomerization, typically dimerization, which juxtaposes the cytoplasmic tyrosine kinase domains [3].
Are receptor tyrosine kinase G proteins?
Receptor tyrosine kinases (RTKs) and trimeric G proteins are 2 such major signaling hubs in eukaryotes. … The molecular mechanisms that govern such non-canonical G protein activation and the relevance of this new paradigm in health and disease is discussed.
What is receptor tyrosine kinase pathway?
Receptor tyrosine kinases (RTKs) are the high-affinity cell surface receptors for many polypeptide growth factors, cytokines, and hormones. … Mutations in receptor tyrosine kinases lead to activation of a series of signalling cascades which have numerous effects on protein expression.
What type of protein is tyrosine kinase?
Protein tyrosine kinase 7 (PTK7) is a single-pass transmembrane protein containing extracellular immunoglobulin domains and an intracellular tyrosine kinase homology domain, although the kinase domain lacks the DFG triplet motif (Lu et al., 2004b).
How do non receptor tyrosine kinases work?
Non-receptor tyrosine kinases are a subgroup of protein family tyrosine kinases, enzymes that can transfer the phosphate group from ATP to a tyrosine residue of a protein (phosphorylation). These enzymes regulate many cellular functions by switching on or switching off other enzymes in a cell.
What is EGFR tyrosine kinase inhibitors?
A substance that blocks the activity of a protein called epidermal growth factor receptor (EGFR). EGFR is found on the surface of some normal cells and is involved in cell growth.
Is EGFR a proto oncogene?
Approximately two decades ago, the epidermal growth factor receptor (EGFR) was discovered to be the proto-oncogene of the mutant, constitutively active oncogenic v-erbB tyrosine kinase, which induces avian erythroblastosis.