Atomic NumberSymbolName1HHydrogen2HeHelium3LiLithium4BeBeryllium
What element that has an atomic mass of 1?
Atomic MassName chemical elementSymbol1.0079HydrogenH4.0026HeliumHe6.941LithiumLi9.0122BerylliumBe
What is element 1 in the periodic table?
Forget 10 Downing Street or 1600 Pennsylvania Avenue, the most prestigious address in the universe is number one in the periodic table, hydrogen.
What electron has an atomic number of 1?
Protons, Neutrons, and ElectronsChargeMass (amu)Proton+11Neutron01Electron−10Is an atom 1 element?
An element is a substance that is made entirely from one type of atom. For example, the element hydrogen is made from atoms containing just one proton and one electron.
What equals the atomic number?
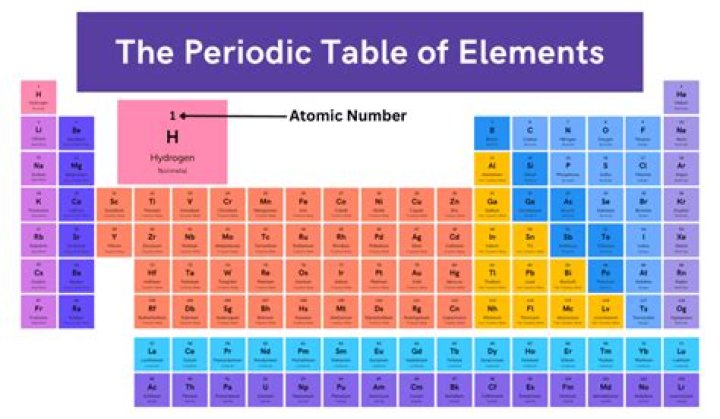
The atomic number equals the charge on the nucleus. It therefore also equals the number of protons in the nucleus and also equals numerically the number of electrons in the neutral atom. The atomic number has the symbol Z.
What is the atomic mass of an element with 1 proton 1 electron and 0 neutron?
For hydrogen, the atomic mass is 1 because there is one proton and no neutrons.
Where is the atomic number of an element?
At the upper left is the atomic number, or number of protons. In the middle is the letter symbol for the element (e.g., H). Below is the relative atomic mass, as calculated for the isotopes found naturally on Earth.Is hydrogen a number 1?
Hydrogen (symbolized H) is one of the elements. Its atomic number is 1 and it is a colorless, tasteless, odorless nonmetallic gas. Hydrogen is highly flammable and is the lightest of all the elements.
What's unusual about Period 1 of the periodic table?Period 1. The first period contains fewer elements than any other, with only two, hydrogen and helium. They therefore do not follow the octet rule, but rather a duplet rule. Chemically, helium behaves like a noble gas, and thus is taken to be part of the group 18 elements.
Article first time published onWhat element has an atomic number of 0?
Neutronium and the periodic table. The term “neutronium” was coined in 1926 by Andreas von Antropoff for a conjectured form of matter made up of neutrons with no protons or electrons, which he placed as the chemical element of atomic number zero at the head of his new version of the periodic table.
What are the 1 to 30 elements?
ATOMIC NUMBERELEMENTATOMIC MASS1Hydrogen1.0082Helium4.00263Lithium6.944Beryllium9.0122
What is an atom Class 9?
The smallest tiny particles of matter which can’t be divided further is called atom, i.e., an atom is the smallest building block of matter. For example: Sodium (Na), Hydrogen (H), Oxygen (O), etc.
What is the smallest thing in the world?
Protons and neutrons can be further broken down: they’re both made up of things called “quarks.” As far as we can tell, quarks can’t be broken down into smaller components, making them the smallest things we know of.
How many atoms are in h2o?
A water molecule has three atoms: two hydrogen (H) atoms and one oxygen (O) atom.
What is the element with 1 proton?
Atomic Number Scientists distinguish between different elements by counting the number of protons in the nucleus (Table 4.5. 1). If an atom has only one proton, we know that it’s a hydrogen atom. An atom with two protons is always a helium atom.
What element has one proton one neutron and one electron?
Deuterium is a hydrogen isotope consisting of one proton, one neutron and one electron. It has major applications in nuclear magnetic resonance studies.
What does a +1 charge mean?
+1 on an atom indicates that,there is a deficiency of one electron. generally protons are positively charged and electrons are negatively charged and are present in equal ration making an atom neutral if electrons are missing positive charge is created and if excess electrons are added the atom is negatively chargeed.
How do you write the atomic number?
- The atomic number or proton number (symbol Z) of a chemical element is the number of protons found in the nucleus of every atom of that element. …
- The sum of the atomic number Z and the number of neutrons N gives the mass number A of an atom.
What are uses for Helium?
Helium is used as an inert-gas atmosphere for welding metals such as aluminum; in rocket propulsion (to pressurize fuel tanks, especially those for liquid hydrogen, because only helium is still a gas at liquid-hydrogen temperature); in meteorology (as a lifting gas for instrument-carrying balloons); in cryogenics (as a …
Why is the Valency of sodium 1?
The electronic configuration of sodium can be written as 2, 8, 1. 2, 8, 1 electrons are distributed in the shells K, L, M respectively. Therefore, valence electron in sodium is 1 and it needs to lose 1 electron from the outermost orbit to attain octet. Hence, the valency of sodium is 1.
What is the atomic number of iron?
In February, we have selected iron, the most abundant element on Earth, with chemical symbol Fe (from the Latin word “ferrum”) and atomic number 26. A neutral iron atom contains 26 protons and 30 neutrons plus 26 electrons in four different shells around the nucleus.
What is the atomic number of this element 2 8 2?
An element has its electron configuration as 2, 8, 2. a. The atomic number of this element is 12.
What is the atomic number of Cl 1?
Element NameChlorineElement SymbolClAtomic Number17
What is the mass of 1 atom of chlorine?
To calculate the relative atomic mass of chlorine, the average mass of one atom of chlorine is found by considering 100 atoms of chlorine. 75.53 of these atoms each have a mass of 35 atomic mass units (AMU), and 24.47 atoms each have a mass of 37 AMU.
What two elements are found in Period 1?
There are only two elements in the first period: hydrogen and helium. We can explain why there is less elements in the first row in modern theories of atomic structure. This is because in quantum physics, this period fills up the 1s orbital.
What do all the elements in Period 1 have in common?
Like PERIODic table.). All of the elements in a period have the same number of atomic orbitals. For example, every element in the top row (the first period) has one orbital for its electrons. All of the elements in the second row (the second period) have two orbitals for their electrons.
How many elements are possible in 1st period?
A period is a horizontal row of the periodic table. There are seven periods in the periodic table, with each one beginning at the far left. A new period begins when a new principal energy level begins filling with electrons. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements.
What are the first 118 elements?
Name of the ElementSymbol of the ElementAtomic NumberArgonAr18PotassiumK19CalciumCa20ScandiumSc21
What is the atomic mass of 1 to 10 elements?
Atomic NumberElementAtomic Mass7Nitrogen14.0078Oxygen15.9999Fluorine18.99810Neon20.18
What is the atomic number of first 20 elements?
Atomic NumberElementSymbol17ChlorineCl18ArgonAr19PotassiumK20CalciumCa