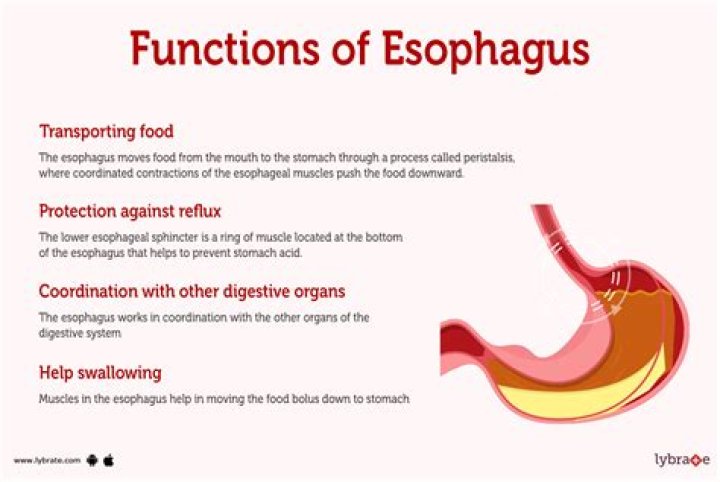
The volume of a single aluminium atom is 1.22×10−23 cm3 . 1 cm3 is equivalent to 0.001 L , or 10−3 L .
What is the volume of a single aluminum atom?
The volume of a single aluminium atom is 1.22×10−23 cm3 . 1 cm3 is equivalent to 0.001 L , or 10−3 L .
How many atoms are there in aluminum?
Finally, to convert the number of moles to atoms, use the fact that 1 mole of aluminium must contain 6.022⋅1023 atoms of aluminium → this is known as Avogadro’s constant.
How do you find the diameter of an aluminum atom?
Set 43πr3 equal to massdensity, so that 43πr3=massdensity, isolate r (radius) and since the diameter is twice the radius, multiply the value for radius by 2. Doing this you get 3.16×10−8 cm.What is the volume of aluminum?
How do you find the volume of aluminium? Determine the volume of the aluminum foil using the density of foil. Volume = mass (g) x density g/cm3 Volume = 4.29 g/ 2.70 g/cm3 Volume= 1.59 cm3 2.
What is the diameter of aluminum?
The diameter of an aluminum atom is approximately 1.2× 10−10m. The diameter of the nucleus of an aluminum atom is approximately 8×10−15m. The density of solid aluminum is 2700kg/m3. a.
What is the mass of 1 atom of Aluminium?
David G. One mole of aluminum atoms has a mass of 26.98 grams, and contains 6.02×1023 atoms.
What is the diameter of an aluminum atom in CM?
An atom of aluminum has a diameter of about 1.2 x 10-8 cm.What is the diameter of each atom?
Everything around us is made up of atoms. An atom is a million times smaller than the thickest human hair. The diameter of an atom ranges from about 0.1 to 0.5 nanometres (1 × 10−10 m to 5 × 10−10 m).
Is aluminum a molecule or atom?The Element Aluminum — Aluminum Atom. Aluminium (or aluminum in North American English) is a chemical element in the periodic table that has the symbol Al and atomic number 13.
Article first time published onHow much is an atom of aluminum?
The cost of one atom of aluminum is $2.06E-25.
What is the mole of aluminum?
One mole of Al atoms has a mass in grams that is numerically equivalent to the atomic mass of aluminum. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g.
How do you find the volume of a single atom?
Calculate the cubic radius of an atom by multiplying the atomic radius by itself three times. For example, if the atomic radius is 5, you would multiply 5 by itself three times, which equals a cubic radius of 125. Use the mathematical formula for the volume of a sphere to calculate the volume of the atom.
What is the formula of volume?
Whereas the basic formula for the area of a rectangular shape is length × width, the basic formula for volume is length × width × height.
What is the mass of 4 atoms of aluminium?
The atomic mass of aluminium is given to be 27u. This means that 1 mole of aluminium atoms has a mass of 27 grams. Thus, the mass of 4 moles of aluminium atoms is 108 grams.
Is the mass of 4 moles of aluminium atoms?
Mass of 4 moles of aluminium atoms = (27 x 4) = 108 g.
What is the mass of 1 mole of aluminium?
Use the periodic table to check the atomic mass, this is the number of grams per mole → 1 mole of Aluminum is 26.982 g ▪ Written as a fraction this is …
How do you find the diameter of a metal atom?
As we know, the diameter is the double of radius of the metal atom. So, the radius is multiplied by $2$ to determine the diameter.
What is the RAM of Aluminium?
A free aluminium atom has a radius of 143 pm.
Are all atoms the same size?
Originally Answered: Are all atoms of same size? No they are not. Each atom has different amounts of protons and neutrons and with increasing amounts of protons and neutrons, the volume of an atom would also increase. For example, Ag (Silver) is 3 times bigger in volume than He (Helium).
How big is an atom in inches?
For example, the diameter of a hydrogen atom is about 10-10 meters, or roughly 4 billionths of an inch (0.000000004 inch).
What is thickness of aluminum foil?
The majority of foil rolls labeled as “Standard Duty” are between . 0004 thick and . 0007 thick. Which at best is slightly over or one half of a mil in thickness.
How many atoms are in a sheet of aluminum foil?
How many atoms thick is aluminum foil? Approximately 193,000 atoms thick.
How do you find the atomic radius?
The radius of an atom can only be found by measuring the distance between the nuclei of two touching atoms, and then halving that distance.
Where is aluminum in the periodic table?
Aluminum is the 13th element on the periodic table. It is located in period 3 and group 13.
Is aluminum molecular or ionic?
Aluminum and chlorine form covalent bonds, not ionic bonds.
How much atoms are in a mole?
The value of the mole is equal to the number of atoms in exactly 12 grams of pure carbon-12. 12.00 g C-12 = 1 mol C-12 atoms = 6.022 × 1023 atoms • The number of particles in 1 mole is called Avogadro’s Number (6.0221421 x 1023).
How many atoms are in a mole calculator?
Avogadro’s number is a very important relationship to remember: 1 mole = 6.022×1023 6.022 × 10 23 atoms, molecules, protons, etc. To convert from moles to atoms, multiply the molar amount by Avogadro’s number.
What color is mole?
A normal mole is usually an evenly colored brown, tan, or black spot on the skin. It can be either flat or raised. It can be round or oval. Moles are generally less than 6 millimeters (about ¼ inch) across (about the width of a pencil eraser).
What is the mole of oxygen?
The mass of oxygen equal to one mole of oxygen is 15.998 grams and the mass of one mole of hydrogen is 1.008 g.
How many grams is a mole?
The mass of one mole of a substance is equal to that substance’s molecular weight. For example, the mean molecular weight of water is 18.015 atomic mass units (amu), so one mole of water weight 18.015 grams.